©
Leandro Castelluccio
(Image taken from: link)
What’s the role of sleep in memory and in the ability of generalization? Does sleep promote learning? Does it have an effect on our ability to generalize knowledge? Does the lack of sleep affect memory consolidation? And what’s the role of the different brain structures on these processes?
The following review is a briefly modified extract (to fit the current essay) from my master’s dissertation, where the role of sleep in learning and generalization is examined.
Sleep and memory consolidation
Sleep has been considered a way in which the brain strengthens memory consolidation after a period of learning (Ekstrand, 1967; Fishbein, 1971; Yaroush, Sullivan, & Ekstrand, 1971). The current literature on the subject abounds with examples ofsleep-related benefits on memory, particularly, the recall of episodic memories has proven to be better after sleep as compared to the retention obtained after a period of wakefulness (Plihal & Born, 1997; Ellenbogen, Hulbert, Jiang, & Stickgold, 2009). Additionally, it has been fostered that sleep could act as a selective tool of which information will be remembered (e.g. Stickgold & Walker, 2013).
According to a recent review by Chatburn, Lushington and Kohler (2014), 27 studies in healthy adults were identified which combined indicate a moderate effect of sleep in facilitating associative memory as tested behaviorally. These authors interpret such findings as supporting reactivation-based models of associative processing. According to them, sleep facilitates behavioral performance on measures of integration of new information into existing schemata and the extraction of rules and gist governing sets of information.
The way in which sleep affects memory and generalization is not well understood. Generalization refers to a broad array of phenomena whereby past experience can be applied to novel settings (Kumaran, 2012). Theliterature shows no consensus onthis subject. Firstly, researchseems to validate the viewthat Rapid Eye Movement sleep (REM) is important for these processes. For example, in a study by Sterpenich et al. (2014), memory cues delivered during REM sleep enhanced subsequent accurate recollections. Coupled with this, researchhas provided ample support forthe “active system consolidation hypothesis”, which states that spontaneous reactivations of recently acquired memories during sleep contribute to their reorganization and integration into long-term memory networks, gradually losing their dependence on the hippocampus and parahippocampal structures and relaying more on the neocortex (Wilson & Mcnaughton, 1994; Skaggs & Mcnaughton, 1996; Peigneuxet al., 2004; Frankland & Bontempi, 2005; Diekelmann & Born, 2010). Secondly, memory consolidation occurs partially via slow wave sleep (SWS)-dependent replay of activity patterns originally evoked during waking (see in Breton & Robertson, 2014). Thirdly, the build-up of general knowledge regarding regular associations appears to involve the coordinated activity of the hippocampus and mediofrontal regions (see in Sweegers, Takashima, Fernández, & Talamini, 2014). A model by O’donnell and Sejnowski (2014) claims that consolidation and generalization of memories occurs in two steps, corresponding to SWS and REM sleep. During the SWS step, neurons in a postsynaptic population are activated through the effects of hippocampal “sharp-wave ripple” inputs. In addition to this, during the following REM step, multiple “weak” activity patterns are sequentially activated in the presynaptic population that propagate to the postsynaptic one, which correspond to cortically-generated REM sleep activity patterns. Such patterns cause potentiation at their activated synapses, inducing memory consolidation.
On the other hand, it is commonly believed that this kind of acquisition of declarative memories is underpinned by gradual changes in neocortical structures. According to McClelland, McNaughton and O’Reilly (1995), the neocortex learns slowly to discover the structure in ensembles of experiences. The hippocampal system permits rapid learning of new items without disrupting this structure, and reinstatement of new memories interleaves them with others to integrate them into structured neocortical memory systems. It is suggested, therefore, that learning complex associative information is initially dependent on encoding processes within the Medial Temporal Lobes (MTLs), specially the hippocampus. Afterwards, spontaneous and repeated activations of hippocampal traces over time allows for neocortical learning in a gradual way (McClelland, McNaughton, & O’Reilly, 1995). Having said that, it has been found that when systems consolidation occurs in the presence of relevant prior knowledge (Bransford, 1979), the assimilation of new paired-associate (PA) memories into existing activated cortical schemas proceeds very rapidly, and the associative encoding of such PAs requires the hippocampus (Tse et al., 2007), but may also involve simultaneous cortical encoding (Tse et al., 2011). Therefore, we could hypothesize that distinct types of learning might involve to a greater degree the hippocampus or the neocortex, as shown by different forms of learning, such as Fast Mapping, which appear to generate this kind of fast cortical changes and acquisition of memories (Atir-Sharon, Gilboa, Hazan, Koilis, & Manevitz, 2015). This assumption could be applied to a blocked and interleaved type of learning. Likewise, it could be suggested that after a period of memory consolidation (for example, an overnight retention interval), hippocampal memory representations would become more reliant on neocortical areas.
There are still discrepancies about the effects of sleep on memory. According to Jurewicz, Cordi, Staudigl and Rasch (2016), there is a presumably accompanied process of decontextualization of the memory trace during consolidation, which means a gradual loss of memory for the learning context. This could be a way in which sleep supports generalization, by a qualitative transformation from perceptually rich and detailed episodic memories to more abstract semantic knowledge. By contrast, current researchseems to validate the viewthat the availability of contextual information generally facilitates memory recall (Jurewicz et al., 2016). As shown in the study by these authors, memory retrieval was significantly improved when the learning context was reinstated, as compared to a different context. Contrary to this, in a study byCairney, Durrant, Musgrove and Lewis (2011), findings suggest a sleep-related reduction in the extent to which context impacts upon retrieval of memories, underpinned by the fact that superior memory after sleep than after wake was observed when learning and retrieval took place in different environmental contexts. This would provide initial support for the possibility that sleep dependent processes may promote a decontextualization of recently formed declarative representations.
Overall, there seems to be no compelling reason to arguethat sleep does not have an effect on memory consolidation, given the large amount of literaturesupporting this fact, yet there is still debate about the extent of this effect, and also how sleep influences generalization and information integration. In connection with this, the formation of conceptual representations, which would require two key computational challenges: integrating information from different sensory modalities and abstracting statistical regularities across exemplars, although they are thought to be facilitated by offline memory consolidation, they seem to be enhanced by wakefulness instead of sleep (Hennies, Lewis, Durrant, Cousins, & Lambon Ralph, 2014). According to the results of these authors’ study, offline memory consolidation facilitated cross-modal category learning, but consolidation across wakefulness, not across sleep, showed this beneficial effect. What’s more, according to a study by Sweegers and Talamini (2014)in which subjects were able to extract complex regularities from multiple associative memories and use them in a generalization task, the performance on this task increased across a 4 hour post-learning period, but no differential effects of sleep and wake states during this interval were observed. Furthermore, sleep benefits on memory seem not to be consistent for all tasks and in all kinds of periods of time. According to Schönauer, Grätsch and Gais (2014), word pair, syllable and motor sequence learning tasks benefit from sleep during the first day after encoding, when compared with day-time or night-time wakefulness. But performance in the wake conditions recovers after another night of sleep. In this study, while sleep deprivation before recall does not impair performance and thus, fatigue cannot adequately explain the lack of long-term effects, it is hypothesized that the hippocampus might serve as a buffer during the retention interval, and consolidation occurs during delayed sleep.
Finally, it is worth noting that there seems to be an interaction between wakefulness and sleep in memory consolidation. In a study by Gregory et al. (2014), in which functional connectivity MRI (fcMRI) was used to examine whether task-induced changes in resting-state connectivity correlate with performance improvement after sleep, it was found that physiological processes immediately after learning correlate with sleep-dependent performance improvement, suggesting that the wakeful resting brain prepares memories of recent experiences for later consolidation during sleep. Both the fcMRI and the sleep control groups showed significant improvement in a motor sequence task performance, while the wake control group did not. In the fcMRI group, increased connectivity in bilateral-motor cortex following the motor task training correlated with the next-day improvement. They hypothesize that the increased connectivity processed the new memories for sleep dependent consolidation. In connection with this, the reactivations by which memory consolidation is assumed to be embodied in synaptic and cellular modifications at brain circuits (by which the memory is initially encoded), occur both during wakefulness and sleep, allowing the distribution of information to additional locales and integrating it into existing knowledge (Dudai, Karni, & Born, 2015). Along similar lines, Bridge and Voss (2014)tested whether across-episode binding preferentially occurs for memory content that is currently “active”, finding that memory for faces was better when tested on the original background scenes, indicating that original episode content was bound with the active condition faces.
Mechanisms by which the hippocampus may support generalization
There is a distinction between two different mechanisms by which the hippocampus may support generalization (Zeithamova, Schlichting, & Preston, 2012): (i) an encoding-based mechanism, which creates overlapping representations that capture higher-order relationships between different items, that may link similar units of information at the point of encoding (e.g. Howard & Kahana, 2002; Shohamy, & Wagner, 2008); and (ii) a retrieval-based mechanism, which effectively computes these relationships at the point of retrieval, through a recurrent mechanism that allows the dynamic interaction of multiple pattern separated episodic codes (Kumaran & McClelland, 2012; DeVito, Kanter, & Eichenbaum, 2010). Generalization in this sense relates to the way in which information is coded or represented in the brain, which is capturing these higher-order relationships of the items that are learned. As Kumaran (2012)points out, the hippocampal replay activity during sleep may under certain conditions be “generalized” in nature, which is reflecting the learning of sequential episodes. This view seems to rest on the assumptionthat there are common overlapping neural codes for related episodes. With this in mind, according to Kumaran (2012), the hippocampus is not thought to be engaged in all forms of generalization, notably in categorization or stimulus generalization. Notwithstanding, it is involved in tasks in which successful performance depends on the ability to appreciate the relationship between discrete items presented in a set of related experiences.
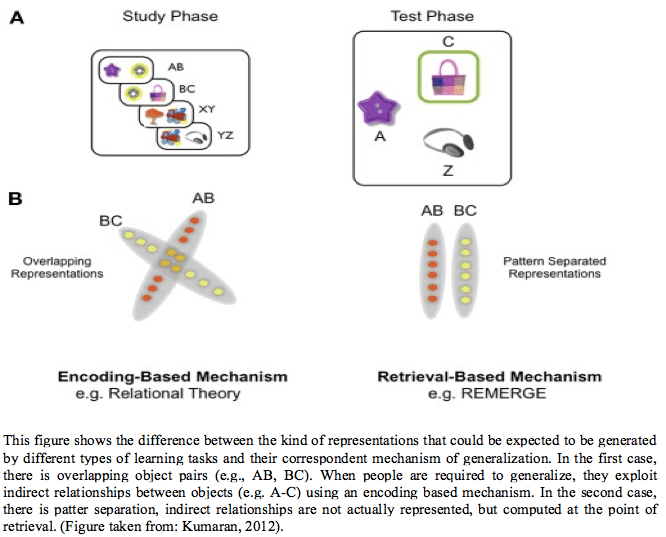
Although the hippocampus has been observed to support both encoding and retrieval- based generalization, it is not clear which conditions might determine which mechanism is primarily used. Nonetheless, it has been recently suggested that strongly learnt association are generalized using an encoding-based mechanism and weakly learnt associations are generalized using a retrieval-based mechanism (Zeithamova, Schlichting, & Preston, 2012). Based on this underlying argument,we could expect that the two kinds of learning (blocked and interleaved) would generate different representations in the brain that would then alter the way in which generalization takes place from these underling representations.
Sleep and generalization
There has been an inconclusive debate about the effects of sleep on generalization, although there is rapidly growing literature on the subject. On the one hand, these two processes seem to be linked, as it is suggested by the fact that sleep affects false memory generation through semantic generalization during the consolidation of the memory trace, or through the recovery function of sleep that affects cognitive control processes of retrieval (Diekelmann, Born, & Wagner, 2010).
There isgrowing supportfor the claimthat sleep has an enhancing effect on generalization (Pace-Schott, Germain, & Milad, 2015; Horváth et al., 2016; Friedrich, Wilhelm, Born, & Friederici, 2015; Earle & Myers, 2015; Batterink, Oudiette, Reber, & Paller, 2014; Javadi, Tolat, & Spiers, 2015; Batterink & Paller, 2015). Despite this, several authors support a positive effect of wake but not sleep on generalization (e.g. Werchan & Gómez, 2014; Hennies et al., 2014). In a middle-ground position, others found no evidence of differences between the wake and sleep conditions on generalization (e.g. Sweegers & Talamini, 2014; Davidson, Carlsson, Jönsson, & Johansson, 2016).
An argument advancedto explain these differences is that many of these studies look at generalization across different age groups. According to Gómez and Edgin (2015), based on the extended trajectory of hippocampal development, it is argued that transitions in the nature of sleep-dependent learning are expected. These authors reviewed a series of studies showing shifts in the nature of sleep-dependent learning across early childhood, with sleep facilitating generalization in infants but enhancing precise memory instead after 18-24 months of age.
One way in which sleep could affect generalization is by consolidating representations. However, Fenn, Margoliash and Nusbaum (2013)for example, directly compared consolidation of rote and generalized learning using a single speech identification task. They showed that training on a large set of novel stimuli resulted in substantial generalized learning, and sleep restored performance that had degraded after 12 waking hours. Rote learning resulted primarily after training on a small set of repeated stimuli. Performance was also degraded after 12 waking hours but was not restored by sleep. Moreover, such performance was significantly worse 24 hours after rote training. This would suggest a functional dissociation between the mechanisms of consolidation for rote and generalized learning.
It is not fully clear whether sleep affects performance in the same way in different types of learning, given the different representations and mechanisms that are involved in each case, nor whether sleep can affect direct or generalized association in the same way.
References
Atir-Sharon, T., Gilboa, A., Hazan, H., Koilis, E., & Manevitz, L. (2015). Decoding the Formation of New Semantics: MVPA Investigation of Rapid Neocortical Plasticity during Associative Encoding through Fast Mapping. 2015, 17.
Batterink, L. J., Oudiette, D., Reber, P. J., & Paller, K. A. (2014). Sleep facilitates learning a new linguistic rule. Neuropsychologia, 65(2014), 169–179. http://doi.org/10.1016/j.neuropsychologia.2014.10.024
Batterink, L. J., & Paller, K. A. (2015). Sleep-based memory processing facilitates grammatical generalization: Evidence from targeted memory reactivation. Brain and Language. http://doi.org/10.1016/j.bandl.2015.09.003
Bransford, J. (1979).Human cognition: Learning, understanding, and remembering. Thomson Brooks/Cole.
Breton, J., & Robertson, E. M. (2014). Flipping the switch: Mechanisms that regulate memory consolidation. Trends in Cognitive Sciences, 18(12), 629–634. http://doi.org/10.1016/j.tics.2014.08.005
Bridge, D. J., & Voss, J. L. (2014). Active retrieval facilitates across-episode binding by modulating the content of memory. Neuropsychologia, 63, 154–164. http://doi.org/10.1016/j.neuropsychologia.2014.08.024
Cairney, S. A., Durrant, S. J., Musgrove, H., & Lewis, P. A. (2011). Sleep and environmental context: Interactive effects for memory. Experimental Brain Research, 214(1), 83–92. http://doi.org/10.1007/s00221-011-2808-7
Chatburn, A., Lushington, K., & Kohler, M. J. (2014). Complex associative memory processing and sleep: A systematic review and meta-analysis of behavioural evidence and underlying EEG mechanisms. Neuroscience and Biobehavioral Reviews, 47, 646–655. http://doi.org/10.1016/j.neubiorev.2014.10.018
Davidson, P., Carlsson, I., Jönsson, P., & Johansson, M. (2016). Sleep and the generalization of fear learning. Journal of Sleep Research, 25(1), 88–95. http://doi.org/10.1111/jsr.12339
DeVito LM, Kanter BR, Eichenbaum H (2010) The hippocampus contributes to memory expression during transitive inference in mice. Hippocampus, 20: 208-217.
Diekelmann, S., Born, J., & Wagner, U. (2010). Sleep enhances false memories depending on general memory performance. Behavioural Brain Research, 208(2), 425–429. http://doi.org/10.1016/j.bbr.2009.12.021
Dudai, Y., Karni, A., & Born, J. (2015). The Consolidation and Transformation of Memory. Neuron,88(1), 20–32. http://doi.org/10.1016/j.neuron.2015.09.004
Earle, F. S., & Myers, E. B. (2015). Overnight consolidation promotes generalization across talkers in the identification of nonnative speech sounds. The Journal of the Acoustical Society of America, 137(January), EL91–EL97. http://doi.org/10.1121/1.4903918
Ekstrand, B. R. (1967). Effect Of Sleep On Memory.Journal of Experimental Psychology, 75(1), 64-72. doi:http://dx.doi.org/10.1037/h0024907
Ellenbogen, J. M., Hulbert, J. C., Jiang, Y., & Stickgold, R. (2009). The sleeping brain’s influence on verbal memory: boosting resistance to interference. PLoS One,4(1), e4117.
Fishbein, W. (1971). Disruptive effects of rapid eye movement sleep deprivation on long-term memory. Physiology & Behavior,6(4), 279-282.
Fenn, K. M., Margoliash, D., & Nusbaum, H. C. (2013). Sleep restores loss of generalized but not rote learning of synthetic speech. Cognition, 128(3), 280–286. http://doi.org/10.1016/j.cognition.2013.04.007
Frankland, P. W., & Bontempi, B. (2005). The organization of recent and remote memories. Nature Reviews Neuroscience, 6(2), 119-130.
Friedrich, M., Wilhelm, I., Born, J., & Friederici, A. D. (2015). Generalization of word meanings during infant sleep. Nature Communications, 6, 6004. http://doi.org/10.1038/ncomms7004
Gregory, M. D., Agam, Y., Selvadurai, C., Nagy, A., Vangel, M., Tucker, M., … Manoach, D. S. (2014). Resting state connectivity immediately following learning correlates with subsequent sleep-dependent enhancement of motor task performance. NeuroImage,102(P2), 666–673. http://doi.org/10.1016/j.neuroimage.2014.08.044
Hennies, N., Lewis, P. A., Durrant, S. J., Cousins, J. N., & Lambon Ralph, M. A. (2014). Time- but not sleep-dependent consolidation promotes the emergence of cross-modal conceptual representations. Neuropsychologia, 63, 116–123. http://doi.org/10.1016/j.neuropsychologia.2014.08.021
Horváth, K., Liu, S., Plunkett, K., Horvath, K., Liu, S., & Plunkett, K. (2016). A daytime nap facilitates generalization of word meanings in young toddlers. Sleep, 39(1), 203–207. http://doi.org/10.5665/sleep.5348
Howard MW, Kahana MJ (2002) A distributed representation of temporal context. Journal of Mathematical Psychology, 46:269-299.
Javadi, A.-H., Tolat, A., & Spiers, H. J. (2015). Sleep enhances a spatially mediated generalization of learned values. Learning & Memory (Cold Spring Harbor, N.Y.), 22(10), 532–6. http://doi.org/10.1101/lm.038828.115
Jurewicz, K., Cordi, M. J., Staudigl, T., & Rasch, B. (2016). No Evidence for Memory Decontextualization across One Night of Sleep. Frontiers in Human Neuroscience, 10(January), 7. http://doi.org/10.3389/fnhum.2016.00007
Kumaran, D. (2012). What representations and computations underpin the contribution of the hippocampus to generalization and inference? Frontiers in Human Neuroscience,6(June), 157. http://doi.org/10.3389/fnhum.2012.00157
Kumaran D, McClelland JL (2012) Generalization through the recurrent interaction of episodic memories: a model of the hippocampal system. Psychological review, 119:573-616.
McClelland, J. L., McNaughton, B. L., & O’Reilly, R. C. (1995). Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychological review, 102(3), 419.
Pace-Schott, E. F., Germain, A., & Milad, M. R. (2015). Effects of Sleep on Memory for Conditioned Fear and Fear Extinction. Psychological Bulletin, 141(4), 835–857. http://doi.org/10.1037/bul0000014
Peigneux, P., Laureys, S., Fuchs, S., Collette, F., Perrin, F., Reggers, J., … & Luxen, A. (2004). Are spatial memories strengthened in the human hippocampus during slow wave sleep?. Neuron, 44(3), 535-545.
Plihal, W., & Born, J. (1997). Effects of early and late nocturnal sleep on declarative and procedural memory. Journal of cognitive neuroscience, 9(4), 534-547.
Schönauer, M., Grätsch, M., & Gais, S. (2014). Evidence for two distinct sleep-related long-term memory consolidation processes. Cortex, 63, 68–78. http://doi.org/10.1016/j.cortex.2014.08.005
Skaggs, W., & Mcnaughton, B. (1996). Replay of Neuronal Firing Sequences in Rat Hippocampus During Sleep Following Spatial Experience. Science,271(5257), 1870-1873.
Shohamy D, Wagner AD (2008) Integrating memories in the human brain: hippocampal-midbrain encoding of overlapping events. Neuron, 60:378-389.
Spencer, R. (2013). Neurophysiological basis of sleep’s function on memory and cognition. ISRN Physiology, 2013.
Sterpenich, V., Schmidt, C., Albouy, G., Matarazzo, L., Vanhaudenhuyse, A., Boveroux, P., … Maquet, P. (2014). Memory reactivation during rapid eye movement sleep promotes its generalization and integration in cortical stores. Sleep, 37(6), 1061–75, 1075A–1075B. http://doi.org/10.5665/sleep.3762
Stickgold, R., & Walker, M. P. (2013). Sleep-dependent memory triage: evolving generalization through selective processing. Nature Neuroscience, 16(2), 139–45. http://doi.org/10.1038/nn.3303
Susanne Diekelmann, & Jan Born. (2010). The memory function of sleep. Nature Reviews Neuroscience,11(2), 114.
Sweegers, C. C. G., Takashima, A., Fernández, G., & Talamini, L. M. (2014). Neural mechanisms supporting the extraction of general knowledge across episodic memories. NeuroImage,87, 138–146. http://doi.org/10.1016/j.neuroimage.2013.10.063
Sweegers, C. C. G., & Talamini, L. M. (2014). Generalization from episodic memories across time: A route for semantic knowledge acquisition. Cortex, 59, 49–61. http://doi.org/10.1016/j.cortex.2014.07.006
Tse, D., Langston, R. F., Kakeyama, M., Bethus, I., Spooner, P. A., Wood, E. R., … & Morris, R. G. (2007). Schemas and memory consolidation. Science, 316(5821), 76-82.
Tse, D., Takeuchi, T., Kakeyama, M., Kajii, Y., Okuno, H., Tohyama, C., … & Morris, R. G. (2011). Schema-dependent gene activation and memory encoding in neocortex. Science,333(6044), 891-895.
Werchan, D. M., & Gómez, R. L. (2014). Wakefulness (Not Sleep) Promotes Generalization of Word Learning in 2.5-Year-Old Children. Child Development, 85(2), 429–436. http://doi.org/10.1111/cdev.12149
Wilson, M., & Mcnaughton, B. (1994). Reactivation of Hippocampal Ensemble Memories During Sleep. Science,265(5172), 676-679.
Yaroush, R., Sullivan, M. J., & Ekstrand, B. R. (1971). Effect of sleep on memory: II. differential effect of the first and second half of the night.Journal of Experimental Psychology, 88(3), 361-366. doi:http://dx.doi.org/10.1037/h0030914
Zeithamova D, Schlichting ML, Preston AR (2012) The hippocampus and inferential reasoning: building memories to navigate future decisions. Frontiers in human neuroscience, 6:70.
Discover more from Sinapticas
Subscribe to get the latest posts sent to your email.

3 thoughts on “Sleep’s effects on memory and generalization”