©
Leandro Castelluccio
(Image taken from: link)
There is a widespread belief that the important thing in many facets of human life is to seek balance and avoid extremes. Food corresponds to one of these aspects, we are often told that healthy nutrition involves eating a bit of everything and avoiding being “dogmatic” or “extremist”, looking for balance between all the food that human beings consume, without restricting certain types of meals and not exceeding daily recommendations about them. This is understood as healthy. Although this topic does not constitute an area that I usually write about, I would like to contribute by showing the overwhelming evidence that exists regarding the impact of the consumption of certain foods on human health since healthy eating is an essential aspect for the well-being of people and the prevention of diseases, and I believe that in this area there are a multitude of misrepresentations of facts, unfounded opinions, bad recommendations and misinformation about widespread empirical evidence that even dates decades back. The belief that the balance is something positive in food consumption is one of these unfounded opinions because within the range of foods that humans consume it is not true that consuming a bit of everything (particularly foods of animal origin) is something better than to select exclusively a particular group of foods (although undoubtedly very varied as in the case of fruits, vegetables and legumes).
Let’s take the case of smoking as an example. The evidence is overwhelming in that the consumption of tobacco is harmful to health. A few years ago this was not the case, not because there was no evidence that smoking tobacco was negative for health, but because despite the fact that such evidence was present for years, in public opinion it was not widespread that smoking was harmful, even to the point that doctors and health professionals did not yet see smoking as a negative fact and even in debates on the subject there would be opposing opinions within the scientific community. But the evidence was there, the studies already existed and they were forceful, but the position was not so marked, the studies were not so well known, where the tobacco industry exerted a lot of pressure and misinformed, all this generating less certainty in general about the subject.
The same case could be considered as to what happens today but with food. This is the opinion for example, of figures like doctor Michael Greger, known promoter of a whole food plant-based diet (fruits, vegetables, legumes and nuts), where the evidence is strong in favor of a diet strictly based on such plant-based foods in terms of promoting good health and protective factors of multiple diseases and contrary in this regard about the consumption of foods of animal origin. We speak particularly of the clear benefits supported by the evidence of a whole food diet, that is, based on complex carbohydrates such as those from legumes and with a high content of vegetables and fruits, proteins of vegetable origin, such as those present in soybeans or quinoa, and unrefined vegetable fats, such as those found in nuts, and we also talk about the evidence that shows the harm to the health of foods such as dairy products, eggs, white and red meats. Although, as we say, the evidence is solid regarding the benefits of the type of diet just mentioned and the harmful effects of the consumption of foods of animal origin, it still persists in the professional health community, and even more so in the media and public opinion in general, that these foods of animal origin are not bad, as long as there is no excess in their consumption, or not even that, because people promote the consumption of such foods as something healthy, such as consuming dairy for its contribution of calcium and because it is a very complete food (in terms of its protein profile), or consuming eggs for the same reasons (up to one a day would not be harmful it is claimed), also fish for its contribution of omega 3 fatty acids, whose intake in the general diet of western countries is low, or consuming chicken because it is considered a white meat with less saturated fat, or even red meat, for its contribution of iron. However, the consumption of these foods is proven to be harmful, even in low quantities, as we will see across this essay. We could say that the status of these foods is equivalent to that of tobacco, in that there is no level of consumption that implies zero risk to human health. What does this mean concretely? That we cannot affirm that smoking one or two cigarettes a day is a safe level of tobacco use for health, we cannot say that consuming one or two cigarettes once or twice a week is also a safe level, that is, there is no safe level of tobacco consumption, any level of consumption puts one’s health at greater risk, although the percentage of increase is slight according to the level of highest consumption (and although it is debated whether consuming a cigarette once every several months generates any risk) .
This is what has yet to be widely disseminated regarding the status of foods of animal origin, but the evidence supports the same fact that there is no safe level of consumption of them, be it dairy products, eggs, white or red meats. Something similar happens in the diet with the consumption of alcohol. It has been popularized that the moderate consumption of it, like a glass of red wine per day, can even be beneficial for health, a fact that is also proven wrong, because in this case there is no safe level of consumption and yet it is promoted the moderate consumption as a healthy habit. As we will see in the next development, any consumption, however minimal, of animal-based foods and alcoholic beverages, increases the risk of suffering from different types of diseases.
Before one thinks that a complete plant-based diet is not appropriate for optimal nutrition, I cite here the position of the US Academy of Nutrition and Dietetics on vegetarian diets (including vegan diets):
“It is the position of the Academy of Nutrition and Dietetics that appropriately planned vegetarian, including vegan, diets are healthful, nutritionally adequate, and may provide health benefits for the prevention and treatment of certain diseases. These diets are appropriate for all stages of the life cycle, including pregnancy, lactation, infancy, childhood, adolescence, older adulthood, and for athletes. Plant-based diets are more environmentally sustainable than diets rich in animal products because they use fewer natural resources and are associated with much less environmental damage. Vegetarians and vegans are at reduced risk of certain health conditions, including ischemic heart disease, type 2 diabetes, hypertension, certain types of cancer, and obesity. Low intake of saturated fat and high intakes of vegetables, fruits, whole grains, legumes, soy products, nuts, and seeds (all rich in fiber and phytochemicals) are characteristics of vegetarian and vegan diets that produce lower total and low-density lipoprotein cholesterol levels and better serum glucose control. These factors contribute to reduction of chronic disease. Vegans need reliable sources of vitamin B-12, such as fortified foods or supplements” (Melina, Craig, & Levin, 2016, page 1970). The paper also mentions two common concerns about vegetarian diets: iron and calcium intake, indicating that vegetarians generally consume as much iron as, or slightly more than omnivores, while having lower iron stores than non-vegetarians, but they add that lower serum ferritin levels may be an advantage because elevated serum ferritin levels have independently been associated with the risk of developing metabolic syndrome. In the case of calcium, intakes of lacto-ovo-vegetarians for example, typically meet or exceed calcium recommendations, while calcium intakes of vegans vary widely and sometimes fall below recommendations. This is why nutrition planning is important, but overall vegans can perfectly achieve optimal nutrition.
There are many myths regarding diet and nutrition and it goes beyond the scope of this essay to cover even some of them, but as an example I would like to mention two of them: that moderate alcohol consumption is healthier as no consumption, and the “French paradox” myth, the notion that rates of several diseases are lower in France despite their diet, which contains a lot of animal products. You can se the following videos by doctor Greger explaining these two issues: Do Any Benefits of Alcohol Outweigh the Risks?–What Explains the French Paradox?
Studies are loaded with statistical analyses of various kinds and in the universe of scientific research it is not uncommon to find contradictory or not replicable results between studies, or often we find different levels of significant effects, or one study showing an effect another showing no effect. This is due to several reasons, some related to the designs of the studies, other to the number of subjects, the natural margins of error, among other variables. The important thing is to keep a sense of perspective and cover multiple studies, seeing the overall picture that emerges so as to pay attention to the general tendency that the data shows. So in the case of our subject, the general tendency in the research, despite what some particular studies found, is to see a clear picture showing adverse effects to human health of consuming animal products. An example of this, is the study by Key et al. (2009), which shows that despite being the overall cancer incidence rates of both the vegetarians and the non-vegetarians in their study low compared with national rates, the incidence of all cancers combined was lower among vegetarians than among meat eaters, but they also get an effect where the incidence of colorectal cancer was higher in vegetarians than in meat eaters. This last finding could be explained by many factors, although to do so is not the main point here, but to simply show that exceptions or contradictory data emerges, but the overall tendency goes in the direction of showing these adverse effects for human health.
Animal consumption does not have to be large in order to see an effect, and this goes with the idea that probably there is no safe level of animal food intake, and each small increase in consumption generates greater impact on health. As Snowdon and Phillips (1985) indicate, even 1-2 servings of meat per week for example seem to increase diabetes risk (you can find more information about this in the following Harvard Magazine link: A Diabetes Link to Meat).There are problems, for example, when trying to set tolerable upper intake levels (ULs) for trans fat, saturated fat and cholesterol. Following Trumbo and Shimakawa (2011), these ULsas set by the Institute of Medicine (IOM) are important, in part because they are used for estimating the percentage of the population at potential risk of adverse effects from excessive nutrient intake. This institution did not set ULs for trans fat, saturated fat and cholesterol because any intake level above 0% of energy increased LDL cholesterol concentration for example, an element linked to Cardiovascular Disease (CD). The results of their analysis confirm the limitations of the risk assessment model for setting ULs because of its inability to identify a UL for food components such as cholesterol, that lack an intake threshold associated with increased chronic disease risk.
In “Beyond Meatless, the Health Effects of Vegan Diets: Findings from the Adventist Cohorts”, authors Le and Sabaté (2014) conduct an extensive analysis on one of the healthiest living populations in the world: the North America’s Adventist, a population with one of the highest, if not the highest, rate of longevity in the world. In the study, the authors go beyond the meat versus non-meat eaters, and study the impact on health of the reduction in animal consumption, and so they compare vegans (who do not consume any animal product, including dairy and eggs) and vegetarians (those who allow dairy and/or eggs, and even some fish).
Following Le and Sabaté (2014) vegetarians and vegans represent only 5% and 2% respectively of the US population. They summarized available evidence from three prospective cohorts of Adventists in North America: Adventist Mortality Study, Adventist Health Study, and Adventist Health Study-2. Non-vegetarian diets were compared to vegetarian dietary patterns (i.e., vegan and lacto-ovo-vegetarian) on selected health outcomes. In general, vegetarian diets confer protection against cardiovascular diseases, cardiometabolic risk factors, some cancers and total mortality.
But compared to lacto-ovo-vegetarian diets, vegan diets seem to offer additional protection for obesity, hypertension, type-2 diabetes, and cardiovascular mortality, where males seem to experience greater health benefits than females. As we observed earlier, this study seems to confirm the findings that 1-2 servings of meat per week is linked with increased diabetes risk. This might be due to increased obesity experienced by meat eaters and omnivores in general, being obesity a risk factor for diabetes. But as we will se, it is probably not so that obesity is the cause of diabetes but rather animal consumption drives increased weight, along with diabetes and heart disease for example, which is closely linked with saturated fat and cholesterol intake. There are additional studies that show this effect of vegan diets on diabetes risk. Barnard et al. (2009) for example, compared the effects of a low-fat vegan diet and conventional diabetes diet recommendations on glycemia, weight, and plasma lipids. Both diets were associated with sustained reductions in weight and plasma lipid concentrations. However, in an analysis controlling for medication changes, a low-fat vegan diet appeared to improve glycemia and plasma lipids more than did conventional diabetes diet recommendations. In an earlier paper, again Barnard et al. (2006) concluded that both a low-fat vegan diet and a diet based on ADA guidelines improved glycemic and lipid control in type 2 diabetic patients but these improvements were greater with a low-fat vegan diet. Goff, Bell, So, Dornhorst and Frost (2005), on the other hand, indicate that vegans have a food intake and a biochemical profile that will be expected to be cardio-protective, with lower intramyocellular lipid accumulation and beta-cell protective, which in turn impacts positively on insulin resistance. Additionally, Pan et al. (2013) indicate that increasing red meat consumption over time is associated with an elevated subsequent risk of type 2 diabetes, and the association is partly mediated by body weight. Their results add further evidence that limiting red meat consumption over time confers benefits for type 2 diabetes prevention.
Overall across literature, we can see that vegetarians have lower incidence of several diseases than meat eaters, but what the extensive study by Le and Sabaté (2014) shows is that when vegetarians are compared with vegans, the incidence is even lower. As the following tables show, the additional reduction in animal product consumption as in a complete vegan diet confers even grater decreased levels of disease, considering cardiometabolic-related factors, all cancer and site-specific cancers and all-cause and cause-specific mortality, which strongly indicates that dairy and egg consumption also has a negative impact on health. A vegetarian diet from a health perspective although being better than an omnivore diet, is not as healthy as a vegan one:


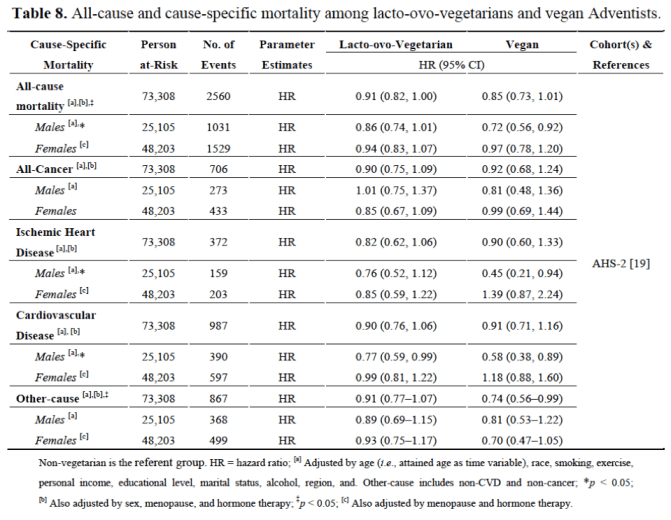
Tables taken from: Le and Sabaté (2014)
So in the Adventist cohorts, both lacto-ovo-vegetarians and vegans have reduced risk for hypertension, type 2 diabetes, and obesity, however, vegans experienced greater risks reduction for those diseases. Similarly, both lacto-ovo-vegetarian males and vegan male have lower mortality from cardiovascular disease compared to non-vegetarians, but risk reduction is greater among vegans. According to the authors, several proposed mechanisms may explain the lower risks for some diseases exhibited by vegans compared to lacto-ovo-vegetarians. As we mentioned earlier, one is the fact that obesity is a major risk factor for CVD and cancer, and as the study shows, significantly lower mean BMI is observed in vegans compared to vegetarians, this may be an important factor for reducing risks in these diseases. As we can see in Spencer, Appleby, Davey and Key (2003), when we compare diet and body mass index in meat- eaters, fish-eaters, vegetarians and vegans, on average, vegans have lower BMI:
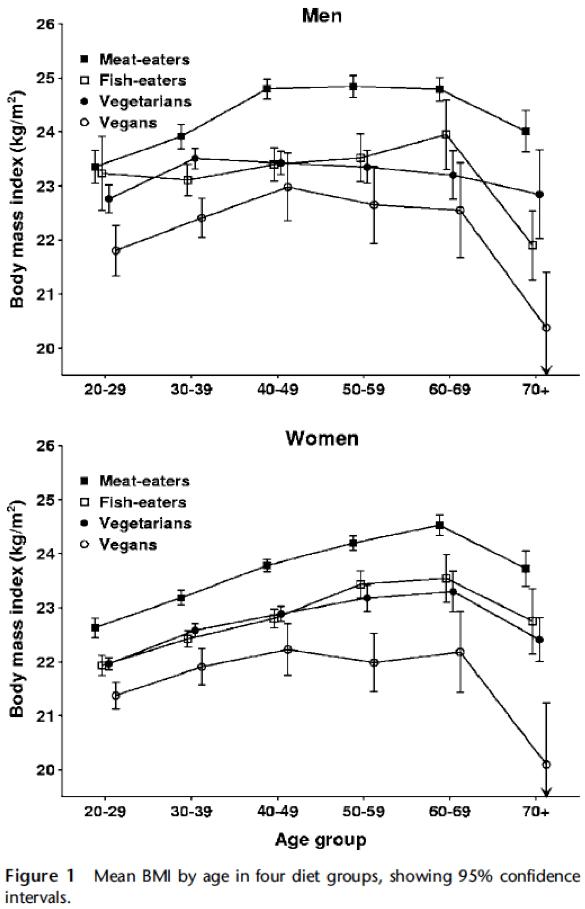

Figure and table taken from: Spencer, Appleby, Davey and Key (2003)
As the data shows, age-adjusted mean BMI was significantly different between the four diet groups, being highest in the meat-eaters (24.41 kg/m2 in men, 23.52 kg/m2 in women) and lowest in the vegans (22.49 kg/m2 in men, 21.98 kg/m2 in women). Fish- eaters and vegetarians had similar, intermediate mean BMI. Differences in lifestyle factors including smoking, physical activity and education level accounted for less than 5% of the difference in mean age-adjusted BMI between meat-eaters and vegans, whereas differences in macronutrient intake accounted for about half of the difference, specifically high protein (as percent energy) and low fiber intakes were the dietary factors most strongly and consistently associated with increasing BMI both between and within the diet groups. These results put meat eaters at a higher risk for obesity and being overweight in general. As we can see in Wright, Wilson, Smith, Duncan and McHugh (2017), who conducted a randomized controlled trial using a whole food plant-based diet (WFPB), at6 months of eating whole plant-based foods, a group of people had a mean BMI reduction greater than when compared with normal care. Mean cholesterol reduction was greater with the WFPB. This program led to significant improvements in BMI, cholesterol and other risk factors, and for the authors, this research has achieved greater weight loss at 6 and 12 months than any other trial that does not limit energy intake or mandate regular exercise, showing how powerful just eating a whole plant-based diet can be in reducing excessive weight.
Obesity is such a major factor in disease that its impact in cancers associated with overweight and obesity make up 40 percent of cancers diagnosed in the United States (you can see this information in the following link: Obesity and Cancer).
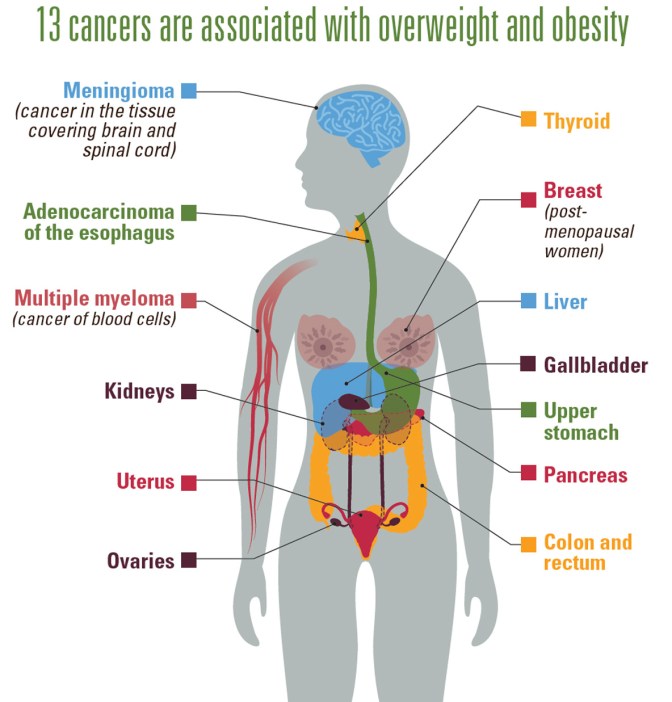
Image taken from: https://www.cdc.gov/media/releases/2017/p1003-vs-cancer-obesity.html
As Wang and Beydoun (2009) point out, meats are high in energy and fat content, and thus may be associated with higher risk of obesity. Considerable differences existed in meat consumption (MC) across sociodemographic groups among US adults, but generally those who consumed more meat had a much higher daily total energy intake, for example, those in the upper versus bottom quintiles consumed around 700 more kcal day(P<0.05). Regression models showed consistent positive associations between MC and BMI, waist circumference, obesity and central obesity. So the study shows results about US national cross-sectional data that make a positive association between MC and risk for obesity and central obesity.
In addition to the aforementioned Adventist study, as we can see in Dinu, Abbate, Gensini, Casini and Sofi (2017), who conducted a comprehensive meta-analysis, a significant protective effect is seen of a vegetarian diet regarding the incidence and/or mortality from ischemic heart disease (-25%) and additionally, lower incidence from total cancer (-8%). While a vegan diet conferred a significant reduced risk (-15%) of incidence from total cancer.
We cannot forget that meat and particularly processed meat have been recently categorized by the World Health Organization (WHO)as type 2 and type 1 carcinogenic, respectively. The following extract is taken from the WHO website about this issue (link):
_______________________________________________________
7. Red meat was classified as Group 2A, probably carcinogenic to humans. What does this mean exactly?
In the case of red meat, the classification is based on limited evidence from epidemiological studies showing positive associations between eating red meat and developing colorectal cancer as well as strong mechanistic evidence.
Limited evidence means that a positive association has been observed between exposure to the agent and cancer but that other explanations for the observations (technically termed chance, bias, or confounding) could not be ruled out.
8. Processed meat was classified as Group 1, carcinogenic to humans. What does this mean?
This category is used when there is sufficient evidence of carcinogenicity in humans. In other words, there is convincing evidence that the agent causes cancer. The evaluation is usually based on epidemiological studies showing the development of cancer in exposed humans.
In the case of processed meat, this classification is based on sufficient evidence from epidemiological studies that eating processed meat causes colorectal cancer.
9. Processed meat was classified as carcinogenic to humans (Group 1). Tobacco smoking and asbestos are also both classified as carcinogenic to humans (Group 1). Does it mean that consumption of processed meat is as carcinogenic as tobacco smoking and asbestos?
No, processed meat has been classified in the same category as causes of cancer such as tobacco smoking and asbestos (IARC Group 1, carcinogenic to humans), but this does NOT mean that they are all equally dangerous. The IARC classifications describe the strength of the scientific evidence about an agent being a cause of cancer, rather than assessing the level of risk.
10. What types of cancers are linked or associated with eating red meat?
The strongest, but still limited, evidence for an association with eating red meat is for colorectal cancer. There is also evidence of links with pancreatic cancer and prostate cancer.
11. What types of cancers are linked or associated with eating processed meat?
The IARC Working Group concluded that eating processed meat causes colorectal cancer. An association with stomach cancer was also seen, but the evidence is not conclusive.
_______________________________________________________
Anderson et al. (2018), for example, found a link between processed meat consumption and breast cancer.As they show in their study, the risk was increased in the highest tertile (>9 g/day) of processed meat consumption. On meta-analysis, processed meat consumption was associated with overall (relative risk [RR] 1.06, 95% CI 1.01–1.11) and post-menopausal (RR 1.09, 95% CI 1.03–1.15), but not pre-menopausal (RR 0.99, 95% CI 0.88–1.10), breast cancer.
In “Mortality from different causes associated with meat, heme iron, nitrates, and nitrites in the NIH-AARP Diet and Health Study: population based cohort study”, authors Etemadi et al. (2017) report an increase in all-causes mortality and death with meat consumption both processed and unprocessed.
Heme iron present in red meat might be associated, among other factors, as we will see, in colon carcinogenesis. Following Bastide et al. (2015), epidemiology shows that red and processed meat intake is associated with an increased risk of colorectal cancer. Heme iron, heterocyclic amines, and endogenous N-nitroso compounds (NOC) are proposed to explain this effect, but their relative contribution is unknown. In their study, aimed at determining, at nutritional doses, which is the main factor involved, heme iron increased the number of preneoplastic lesions, but dietary heterocyclic amines and NOC had no effect on carcinogenesis in rats. Dietary hemoglobin increased tumor load in Min mice. Genotoxicity was also observed in colon mucosa of mice given hemoglobin. The results highlight the role of heme iron in the promotion of colon cancer by red meat. The authors also suggest that heme iron could initiate carcinogenesis through lipid peroxidation.
The incredible impact of a whole food plant-based diet on cancer is seen in Barnard, Hong Gonzalez, Liva and Ngo (2006), who investigated the effects of a diet and exercise intervention on known breast cancer (BCa) risk factors, including estrogen, obesity, insulin, and insulin-like growth factor-I (IGF-I), in overweight/obese, postmenopausal women. Using the subjects’ pre and post intervention serum in vitro, serum-stimulated growth and apoptosis of three estrogen receptor-positive BCa cell lines were studied. Women where placed on a low-fat (10-15% kcal), high-fiber (30-40 g per 1,000 kcal/day) diet and attended daily exercise classes for 2 weeks. Serum estradiol was reduced in the women on hormone treatment (HT; n = 28 as well as those not on HT (n = 10). Additionally, serum insulin and IGF-I were significantly reduced in all women, whereas IGF bindingprotein-1 was increased significantly (which has a positive effect in reducing IGF-I). Results show that in vitro growth of the BCa cell lines was reduced by 6.6% for the MCF-7 cells, 9.9% for the ZR-75-1 cells, and 18.5% for the T-47D cells. Apoptosis was increased by 20% in the ZR-75-1 cells, 23% in the MCF-7 cells, and 30% in the T-47D cells (n = 12). These results show that a very-low-fat, high-fiber diet combined with daily exercise results in major reductions in risk factors for BCa while subjects remained overweight/obese, while these in vivo serum changes slowed the growth and induced apoptosis in serum-stimulated BCa cell lines in vitro.

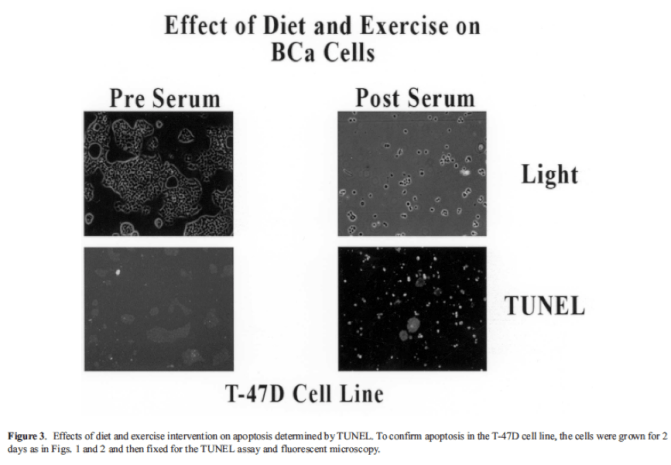
Figures taken from Barnard et al. (2006)
Barnard, Hong and Liva (2005) had indicated before that a very low-fat, high-fiber diet combined with daily exercise results in major reductions in recognized risk factors for BCa. These in vivo changes in serum factors slowed the growth rate and induced apoptosis in BCa cell lines in vitro. What the previous results show is that the blood of women under a plant based diet combined with daily exercise is much more hostile to cancer cells than those under a normal diet which includes animal products, significantly reducing serum BCa risk factors including estradiol, insulin, and IGF-I in overweight/obese, postmenopausal women. These changes were found in both women on hormone treatment and those not on hormone treatment. The results suggest a significant reduction in the risk for BCa or for BCa recurrence. As you can see in the following video by doctor Greger: link, there was an additional study analyzing the relative impact of exercise and diet on cancer cells’ apoptosis, showing that even tenuous and long term exercise (although having a positive effect on this outcome) is not as powerful as a plant based vegan diet on stopping cancer cell growth and inducing apoptosis. This does not mean that we should favor one thing above the other. Following Moore et al. (2016), leisure-time physical activity has been associated with lower risk of heart disease and all-cause mortality, and as the authors mention, with lower risks of many cancer types. Most of these associations were evident regardless of body size or smoking history, supporting broad generalizability of the findings. The best approach probably implies combining the two, a plant-based diet plus physical exercise.
So one of the driving factors that might explain why a vegan diet confers this protective effect against cancer is IGF-I (insulin-like growth factor-I), which seems to be linked with every stage of cancer, from mutation, growth and dissemination (metastasis) – see in previous link of doctor Greger’s video. It appears that animal protein might induce increased levels of IGF-I, while in vegan diet this does not happen, in addition that a vegan diet promotes factors that reduce de levels of IGF-1. As Allen et al. (2002) point out, vegans have lower IGF-1 levels. In their cross-sectional study, aimed at determining whether a plant-based (vegan) diet is associated with a lower circulating level of insulin-like growth factor I (IGF-I) compared with a meat-eating or lacto-ovo-vegetarian diet among 292 British women, ages 20–70 years, the mean serum IGF-I concentration was 13% lower in 92 vegan women compared with 99 meat-eaters and 101 vegetarians . Also, the mean concentrations of both serum IGF-binding protein, IGFBP-1 and IGFBP-2, were 20–40% higher in vegan women compared with meat-eaters and vegetarians (these factors decrease the level of IGF-I). There were no significant differences in IGFBP-3, C-peptide, or sex hormone-binding globulin concentrations between the diet groups. It was shown that the intake of protein rich in essential amino acids was positively associated with serum IGF-I and explained most of the differences in IGF-I concentration between the diet groups. These data suggest that a plant-based diet is associated with lower circulating levels of total IGF-I and higher levels of IGFBP-1 and IGFBP-2, and it indicates that if protein intake correlates high with this elevated levels of IGF-I, it is animal protein the key factor, not plant-based protein. As Appleton and Campbell (1983) found in their experiments, high protein diets (containing Casein found in milk) increased tendency for tumor development.Following Allen et al. (2002), prospective epidemiological studies have shown that elevated levels of IGF-I, as absolute concentrations or relative to levels of IGFBP-3, are associated with an increased risk of breast cancer in premenopausal women for example. IGF-I might play a key role in prostate cancer also. Allen, Appleby, Davey and Key (2000)studied this possible link in the largest to that date study to investigate differences in serum hormone concentrations between meat-eaters, vegetarians and vegans showing a significant 9% lower IGF-I concentration among vegan men compared to meat-eaters and vegetarians as well. Following the authors, IGF-I may play an important role in the aetiology of prostate cancer via its ability to interact with androgens to stimulate prostatic cell growth. In a study cited by their paper (Chan, J. M., Stampfer, M. J., Giovannucci, E., Gann, P. H., Ma, J., Wilkinson, P., … & Pollak, M. (1998)). Plasma insulin-like growth factor-I and prostate cancer risk: a prospective study. Science, 279(5350), 563-566.), men who subsequently developed prostate cancer had 8% higher serum IGF-I concentrations than men who remained healthy, suggesting that the 9% difference they observed could be large enough to significantly alter prostate cancer risk.
Additionally, Allen et al. (2000) found that vegans had higher testosterone levels than vegetarians and meat-eaters, but this was offset by higher sex hormone binding globulin, and there were no differences between diet groups in free testosterone, androstanediol glucuronide or luteinizing hormone. In this regard, it appears that it is common to think that eating meat is more manly or masculine than eating just plant-based foods. In “Meat, morals, and masculinity”, authors Ruby and Heine (2011) indicatethat much research has demonstrated that people perceive consumers of “good”-low-fat foods as more moral, intelligent, and attractive, and perceive consumers of “bad”-high-fat foods as less intelligent, less moral, and less attractive, but little research has contrasted perceptions of omnivores and vegetarians, particularly with respect to morality and gender characteristics. In two between-subject studies, these authors investigated people’s perceptions of others who follow omnivorous and vegetarian diets, controlling for the perceived healthiness of the diets in question. In both studies, omnivorous and vegetarian participants rated vegetarians as more virtuous but also less masculine than omnivores. It is also worth noting that males eat more meat than females, as in the case of the US population (you can check this information in the following link):
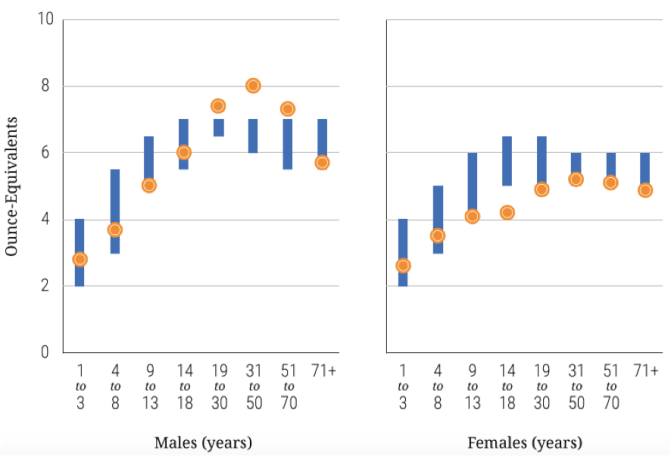
Image taken from: https://health.gov/dietaryguidelines/2015/guidelines/chapter-2/a-closer-look-at-current-intakes-and-recommended-shifts/ – figure-2-6
Going back to IGF-I, as Guevara-Aguirre et al. (2011) indicate, life span extending mutations in growth signaling pathways protect against age-dependent DNA damage in yeast and decrease insulin resistance and cancer in mice and so to test their effect in humans they monitored for 22 years Ecuadorian subjects with mutations in the growth hormone receptor gene leading to severe growth hormone receptor (GHR) and IGF-I deficiencies and combined this information with surveys to identify the cause and age of death for subjects who died before this period. The individuals with GHR deficiency (GHRD) exhibited only one non-lethal malignancy and no cases of diabetes, in contrast to 17% cancer and 5% diabetes prevalence in the controls.Additionally, a considerable amount of evidence is consistent with the proposition that systemic IGF-I activity acts as pacesetter in the aging process (McCarty, 2003). As the author indicates, reduction in IGF-I activity is the common characteristic of rodents whose maximal lifespan has been increased by a wide range of genetic or dietary measures, including caloric restriction. The lifespans of breeds of dogs and strains of rats tend to be inversely proportional to their mature weight and IGF-I levels. The link between IGF-I and aging appears to be evolutionarily conserved; in worms and flies, lifespan is increased by reduction offunction mutations in signaling intermediates homologous to those which mediate insulin/IGF-I activity in mammals. The fact that an increase in IGF-I activity plays a key role in the induction of sexual maturity is consistent with a broader role for-IGF-I in aging regulation. And so, down-regulation of IGF-I activity could slow aging in humans, while a range of practical measures for achieving this may be at hand, including a low-fat, whole-food, vegan diet, exercise training, soluble fiber, insulin sensitizers, appetite suppressants, and agents such as flax lignans, oral estrogen, or tamoxifen that decrease hepatic synthesis of IGF-I (McCarty, 2003). As the author points out, many of these measures would also be expected to decrease risk for common age-related diseases.
Animal consumption not only promotes cancer, but also Cardiobascular Disease. In this regard, a whole food plant-based diet promotes healthy heart and circulatory function. In “A way to reverse CAD”, Esselstyn, Gendy, Doyle, Golubic and Roizen (2014), show that a plant-based nutrition achieved coronary artery disease (CAD) arrest and reversal in a small study, indicating that CAD can potentially be reversed by a strict plant-based diet. In their follow-up study 198 consecutive patients volunteers received counseling to convert from a usual diet to plant-based nutrition. Of the 198 patients with CVD, 177 (89%) were adherent. Major cardiac events judged to be recurrent disease totaled one stroke in the adherent cardiovascular participants, a recurrent event rate of .6%. Thirteen of 21 (62%) non-adherent participants experienced adverse events. Most of the volunteer patients with CVD responded to intensive counseling, and those who sustained plant-based nutrition for a mean of 3.7 years experienced a low rate of subsequent cardiac events.
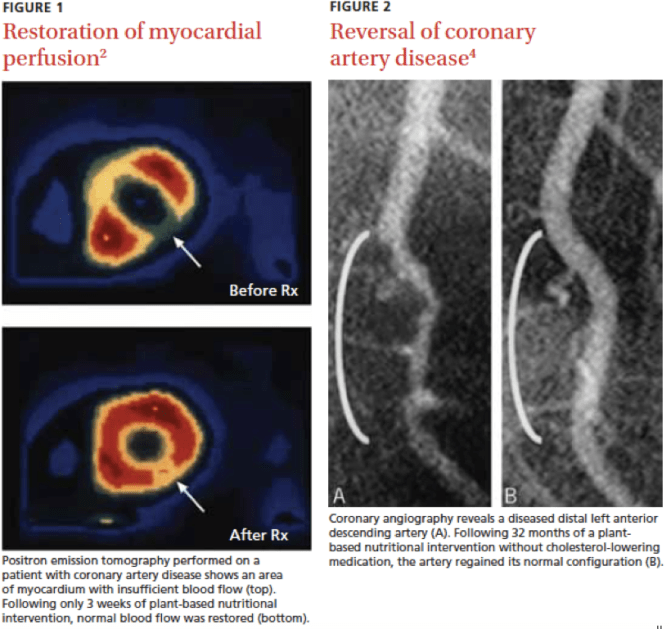
Image: Esselstyn CB Jr (2007). Prevent and Reverse Heart Disease. New York, New York: Penguin Group.
The effects of animal products in heart disease, specially the contribution of saturated fat and cholesterol seem to be increasingly debated, where high fat and protein and low carb diets are rising as “healthy” dietary habits. As Spence, Jenkins and Davignon (2012) point out, increasingly the potential harm from high cholesterol intake, and specifically from egg yolks, is considered insignificant. These authors assessed total plaque area (TPA) in patients attending Canadian vascular prevention clinics to determine if the atherosclerosis burden, as a marker of arterial damage, was related to egg intake. In order to provide perspective on the magnitude of the effect caused by egg consumption, they also analyzed the effect of smoking (pack-years). Data were available in 1262 patients. Results show that carotid plaque area increased linearly with age after age 40, but increased exponentially with pack-years of smoking and with egg-yolk years. Plaque area in patients consuming <2 eggs per week (n = 388) was 125 ± 129 mm2, versus 132 ± 142 mm2 in those consuming 3 or more eggs per week (n = 603); (p < 0.0001 after adjustment for age). In multiple regression, egg-yolk years remained significant after adjusting for coronary risk factors. So their findings suggest that regular consumption of egg yolk should be avoided by persons at risk of cardiovascular disease. But what this study also shows, is that eating something often regarded as “healthy” as eggs, even just once a day (some have claimed that one egg a day does not have a negative effect on CVD), is as harmful as smoking. This connection is what has not yet been generally widespread among the public, not so the egg consumption-carotid plaque association, but the fact that animal products, such as eggs or meat, can be as harmful as smoking, in this sense, conceptually, they are like the same thing. Confirming this fact Li, Zhou, Zhou, and Li (2013) assessed the dose–response relationship between egg consumption and the risk of CVD and diabetes finding results that suggests that there is a dose–response positive association between egg consumption and the risk of CVD and diabetes.
In general, it is observed that low-carbohydrate (LC)diets impair flow-mediated dilatation (FMD), LC dietary protocols have been associated with a significant decrease in FMD when compared with their low-fat (LF) counterparts (Schwingshackl & Hoffmann, 2013). Usually, LC diets include mostly animal protein and fat. In a systematic review and meta-analysis of observational studies, authors Noto, Goto, Tsujimoto and Noda (2013) found that LC diets were associated with a significantly higher risk of all-cause mortality, although they did not find a significant effect associated with a risk of CVD mortality and incidence. However, as the authors mention, their analysis is based on limited observational studies and large-scale trials on the complex interactions between LC diets and long-term outcomes are needed. It is important to note that legumes for example (which contain complex carbohydrates), is the most important dietary predictor of survival in older people of different ethnicities according to Darmadi-Blackberry et al. (2004). In their study, in order to identify protective dietary predictors amongst long-lived elderly people (N= 785), the “Food Habits in Later Life” (FHILL) study was undertaken among five cohorts in Japan, Sweden, Greece and Australia. Between 1988 and 1991, baseline data on food intakes were collected. There were 785 participants aged 70 and over that were followed up to seven years. Based on an alternative Cox Proportional Hazard model adjusted to age at enrollment (in 5-year intervals), gender and smoking, the legume food group showed 7-8% reduction in mortality hazard ratio for every 20g increase in daily intake with or without controlling for ethnicity. Other food groups were not found to be consistently significant in predicting survival amongst the FHILL cohorts. Following Brinkworth, Buckley, Noakes, Clifton and Wilson (2009), in their study, over 1 year, there was a favorable effect of an energy-restricted LF diet compared with an isocaloric LC diet on mood state and affect in overweight and obese individuals. Both diets had similar effects on working memory and speed of processing, confirming the beneficial effects of having a diet based on carbohydrates versus high fat and protein diets (like the so called “Paleo diet”).
Another population with the highest rates of centenarian in the world, the Okinawans in Japan, had a diet were the majority of the energy intake came from complex carbohydrates (between 80 and 90%), in which sweet potatoes were the pillar of their diet. As Willcox, Scapagnini and Willcox (2014) point out, the traditional diet in Okinawa is anchored by root vegetables (principally sweet potatoes), green and yellow vegetables, soybean-based foods, and medicinal plants. On the other hand, many characteristics of the traditional Okinawan diet are shared with other healthy dietary patterns, including the traditional Mediterranean diet for example. Overall, these shared dietary patterns refer to high intake of unrefined carbohydrates, moderate protein intake with emphasis on vegetables/legumes, fish, and lean meats as sources, and a healthy fat profile (higher in mono/polyunsaturated fats, lower in saturated fat; rich in omega-3). As the authors mention, the healthy fat intake is likely one mechanism for reducing inflammation, optimizing cholesterol, and other risk factors, as we will address later. Additionally, the lower caloric density of plant-rich diets results in lower caloric intake with concomitant high intake of phytonutrients and antioxidants. Also, other shared features include low glycemic load, less inflammation and oxidative stress, and potential modulation of aging-related biological pathways. This may reduce risk for chronic age-associated diseases and promote healthy aging and longevity. The Okinawans of old were not vegetarians but they were close to this eating pattern, but not by any particular conscious choice. As Willcox et al. (2014) mention, like most Asian populations, the average family simply could not afford meat, nor processed foods such as sugar, salt, cooking oil, or in the case of Okinawa, imported polished white rice. Staple foods were what could grow locally. Since Okinawa consists of a string of subtropical islands with seasonal, quite severe tropical storms, they had two growing seasons, which favored fresh plant foods. Some vegetables grew very well but rice did not. In fact, rice was supplanted in the 1600s by the sweet potato as the main staple, when it was first imported from China. The sweet potato is hardy and can survive severe climates and thus became the main calorie source. Most other plant foods were also grown and consumed locally (Willcox et al., 2014).
Dietary Carbohydrates might have played an important role in human evolution, as Hardy, Brand-Miller, Brown, Thomas and Copeland (2015) propose, plant foods containing high quantities of starch were essential for the evolution of the human phenotype during the Pleistocene. As they mention, although previous studies have highlighted a stone tool-mediated shift from primarily plant-based to primarily meat-based diets as critical in the development of the brain and other human traits, they argue that digestible carbohydrates were also necessary to accommodate the increased metabolic demands of a growing brain. Furthermore, they acknowledge the adaptive role cooking played in improving the digestibility and palatability of key carbohydrates. The authors provide evidence that cooked starch, a source of preformed glucose, greatly increased energy availability to human tissues with high glucose demands, such as the brain, red blood cells, and the developing fetus. As they mention, although uncertainties remain regarding the antiquity of cooking and the origins of salivary amylase gene copy number variation, the hypothesis they present makes a testable prediction that these events are correlated. Overall, there is great probability that we are best adapted to eat a whole food plant-based diet in which cooking plays a major role, instead of an omnivore one. This correlates with the fact that as we approach mostly a plant-based diet, our health and life span increases.
Going back to animal product consumption and CVD, papers like the one by Hyson, Rutledge and Berglund (2003) show that mechanistic studies demonstrate that triglyceride-rich lipoprotein remnants may have adverse effects on endothelium and can penetrate into the subendothelial space. Exchange of core lipids between postprandial lipoproteins and low-density lipoprotein (LDL)/high-density lipoprotein (HDL) is increased during prolonged lipemia, resulting in small, dense LDL particles (the so called “bad cholesterol”) and reduced HDL cholesterol levels. As the authors mention, collectively, the data suggest that assessment and treatment of atherosclerosis should include parameters related to postprandial lipemia, which is affected by fat intake. A great way to reduce this effect is limiting fat consumption, especially from animal sources, while substituting for monounsaturated and polyunsaturated fatty acids like the ones found in vegetable oils.As Lichtenstein et al. (1993) mention, saturated fat is recommended to be limited, but there is debate as to whether the remaining fat in the diet should be relatively high in monounsaturated or polyunsaturated fatty acids. The authors examined this issue by testing the effects of diets meeting the aforementioned guidelines that were enriched in three different vegetable oils on plasma lipids in the fasting and postprandial states in a clinically relevant population. Plasma cholesterol concentrations declined after consumption of diets enriched in all the tested oils, however, the declines were significantly greater for the canola (12%) and corn (13%) than for the olive (7%) oil-enriched diet. The major finding of the study, as the authors mention, is that significant reductions in LDL-C and apoB levels can be achieved in middle-aged and elderly women and men with initial LDL-C levels > 130 mg/dL by reducing dietary saturated fat and cholesterol intake and by incorporating vegetable oils rich in either monounsaturated fatty acids (canola and olive oil) or polyunsaturated fatty acids (corn oil). It is worth mentioning that although differential effects were seen after the consumption of the three different oil-enriched diets in some plasma lipid measures, none of these oils had a significant advantage in terms of altering the overall lipoprotein profile, and olive oil might not be as healthy as other types of oil, like canola oil.
There is a major issue here regarding the omega 6 to omega 3 ratio. Vegetable oils usually consumed in the diet have mostly omega 6, and the high intake if this fatty acid compared to omega 3 is linked to adverse effects on health. As Simopoulos (2002) mentions, several sources of information suggest that human beings evolved on a diet with a ratio of omega-6 to omega-3 essential fatty acids (EFA) of approximately 1 whereas in Western diets the ratio is 15/1-16.7/1. So western diets are deficient in omega-3 fatty acids, and have excessive amounts of omega-6 fatty acids compared with the diet on which human beings evolved and their genetic patterns were established. According to the author, excessive amounts of omega-6 polyunsaturated fatty acids (PUFA) and a very high omega-6/omega-3 ratio, as is found in today’s Western diets, promote the pathogenesis of many diseases, including cardiovascular disease, cancer, and inflammatory and autoimmune diseases, whereas increased intake of omega 3 and a low omega-6/omega-3 ratio seem to exert suppressive effects. The author further notes that in the secondary prevention of cardiovascular disease, a ratio of 4/1 was associated with a 70% decrease in total mortality. A ratio of 2.5/1 reduced rectal cell proliferation in patients with colorectal cancer, whereas a ratio of 4/1 with the same amount of omega-3 PUFA had no effect. The lower omega-6/omega-3 ratio in women with breast cancer was associated with decreased risk. A ratio of 2-3/1 suppressed inflammation in patients with rheumatoid arthritis, and a ratio of 5/1 had a beneficial effect on patients with asthma, whereas a ratio of 10/1 had adverse consequences. So the optimal ratio may vary with the disease under consideration, but the fact is that a lower ratio of omega-6/omega-3 fatty acids is more desirable in reducing the risk of many of the chronic diseases of high prevalence in Western societies, as well as in the developing countries (Simopoulos, 2002).
Inflammation seems to play a mayor role here. For example, we can consider the case of neuroinflammation (Farooqui, Horrocks, & Farooqui, 2007). As these authors mention, neuroinflammation is a hallmark of all major central nervous system diseases. Docosahexaenoic acid (DHA), a type of omega 3, and its lipid mediators prevent neuroinflammation by inhibiting transcription factor NFκB, preventing cytokine secretion, blocking the synthesis of prostaglandins, leukotrienes, and thromboxanes, and modulating leukocyte trafficking. The dietary intake of docosahexaenoic acid offers the possibility of counter‐balancing the harmful effects of high levels of arachnoid acid‐derived pro‐inflammatory lipid mediators.
This goes hand in hand with the inflammation theory of disease. As Hunter (2012) mentions, an increasing body of evidence shows that chronic inflammation causes and advances many common diseases. In these sense a vegan diet offers great anti-inflammatory power, as vegan diets are shown to lower inflammation. Following Sutliffe, Wilson, de Heer, Foster, and Carnot (2015), a tested brief lifestyle intervention, including a vegan diet rich in fresh fruits and vegetables, whole grains and various legumes, nuts and seeds, significantly improved health risk factors and reduced systemic inflammation as measured by circulating CRP (C-reactive protein). The degree of improvement was associated with baseline CRP such that higher levels predicted greater decreases. As the author mention, it is likely that the vegetable and high fiber content of a vegan diet reduces CRP in the presence of obesity. Additionally, those participants who had a vegan diet prior to the intervention had the lowest CRP risk coming into the program.
Also, considering that taking more omega 3 and balancing the omega 6/omega 3 ratio might promote an anti-inflammatory effect, a vegan diet is a great way to achieve that. However, it is usually argued that people can only get adequate amounts of omega 3 through the consumption of fish or fish oil. In the vegetable kingdom, the most common form of omega 3 is alpha-linolenic acid (ALA), but the body also needs the other two forms: eicosapentaenoic acid (EPA) and the aforementioned docosahexaenoic acid (DHA), which are not very common in vegetables and legumes for example. But the body can convert ALA into EPA and DHA, although it is usually argued that the conversion process is not very efficient and only small percentages of ALA get converted into the other two forms. According to Gester (1998) the use of ALA labeled with radioisotopes suggested that with a background diet high in saturated fat conversion to long-chain metabolites is approximately 6% for EPA and 3.8% for DHA. With a diet rich in n-6 PUFA, conversion is reduced by 40 to 50%. This means that high intake of omega 6 reduced the level of conversion of ALA to EPA and DHA. But even if we take into account the above small percentages of conversion, one tablespoon of flaxseed oil for example, has 7.26 grams of omega 3 in the form of ALA (see in:https://ods.od.nih.gov/factsheets/Omega3FattyAcids-HealthProfessional/), so at the above conversion rate, just one tablespoon will give you 435 mg of EPA and 276 mg of DHA. Thus just by adding one or two tablespoons of this oil to the diet is more than enough to not only meet recommendations of omega 3 intake, but to incorporate higher intake of omega 3 as well. Now, let’s compare this with the EPA and DHA content of average fish oil capsules as they are labeled: about 180 mg/g EPA and 120 mg/g DHA, as shown in Yi et al. (2014). Although as the authors show, the actual content might vary in these capsules, one would need to take several pills generally to achieve what two tablespoons of flaxseed oil can offer (this without taking into account other sources of ALA in a vegan diet). Yi et al. (2014) reported finding capsules with actual contents as low as 39.52 mg/g for EPA and 35.14 mg/g for DHA.
So a whole food plant-based diet might also include reducing significantly the use and consumption of refined oils to lower omega 6 intake, and boosting ALA sources instead, like flaxseeds or flaxseeds oil and canola oil.
Going back to diet and CVD, Stamler, Wentworth and Neaton (1986) report that the relationship between serum cholesterol and Coronary Heart Desease is not a threshold one, with increased risk confined to the two highest quintiles, but rather is a continuously graded one that powerfully affects risk for the great majority of middle-aged American men for example. So the more cholesterol we take in the diet the more risk for such disease, there is no such safe level of intake as it recently portrayed in the general opinion about egg consumption form example. Following Levy, Maor, Presser and Aviram (1996), eggs consumption, in addition to its hypercholesterolemic effect, increases plasma and LDL oxidizability, a phenomenon which was shown to enhance the progression of atherosclerosis. The atherogenic properties may contribute to the accelerated atherosclerosis prevalent in populations with high cholesterol intake. Additionally, in a video by doctor Greger (link), it is shown how high levels of HDL might not offer a protective effect against Heart Desease, but instead lower levels of LDL are an important preventive factor. As Voight et al. (2012) report, some genetic mechanisms that raise plasma HDL cholesterol do not seem to lower risk of myocardial infarction. These data challenge the concept that rising of plasma HDL cholesterol will uniformly translate into reductions in risk of myocardial infarction. And a vegetarian diet is associated to lower levels of triglycerides, total cholesteroland LDL as compared to the diet of omnivores, as found by De Biase, Fernandes, Gianini and Duarte (2007).
It is also worth mentioning that Alzheimer’s pathogenesis for example, might have a vascular basis, and as such, high consumption of saturated fat and cholesterol might be a risk factor for developing the disease. As de la Torre (2002) mentions, considerable evidence now indicates that Alzheimer’s disease (AD) is primarily a vascular disorder. According to the author, this conclusion is supported by the following evidence: (1) epidemiologic studies linking vascular risk factors to cerebrovascular pathology that can set in motion metabolic, neurodegenerative, and cognitive changes in Alzheimer brains; (2) evidence that AD and vascular dementia (VaD) share many similar risk factors; (3) evidence that pharmacotherapy that improves cerebrovascular insufficiency also improves AD symptoms; (4) evidence that preclinical detection of potential AD is possible from direct or indirect regional cerebral perfusion measurements; (5) evidence of overlapping clinical symptoms in AD and VaD; (6) evidence of parallel cerebrovascular and neurodegenerative pathology in AD and VaD; (7) evidence that cerebral hypoperfusion can trigger hypometabolic, cognitive, and degenerative changes; and (8) evidence that AD clinical symptoms arise from cerebromicrovascular pathology.
We cannot forget the effects on health of milk and dairy products in general. As Maruyama, Oshima and Ohyama (2010) argue, modern genetically improved dairy cows continue to lactate throughout almost the entire pregnancy, therefore, recent commercial cow’s milk contains large amounts of estrogens and progesterone. The authors found that estrogens in milk were absorbed for men and children, and gonadotropin secretion was suppressed, followed by a decrease in testosterone secretion. Sexual maturation of prepubertal children could be affected by the ordinary intake of cow milk according to the authors. According to Farlow, Xu and Veenstra (2010), on the whole, evidence for an increase in risk for breast cancer through consumption of cow’s milk and dairy products is blurry and partially contradictory and equivocal. Following the authors, one important reason for this situation might be found in exposure timing: exposure during childhood has to be differentiated from intake in adult life when breast tissue has finally differentiated after successful pregnancy. But alterations in animal production have led to significantly increased alimentary uptake of animal steroids and their metabolites and several sources point to a negative effect of milk consumption.Wiley (2011), for example, shows that there is some evidence that greater milk intake is associated with an increased risk of early menarche, or a lower age at menarche, this might affects later development of breast cancer (Apter & VIHKO, 1983). Contrary to this, in a population-based, prospective cohort study in Japan, frequent miso soup and isoflavone consumption (such as those found in soymilk) was associated with a reduced risk of breast cancer (Torfadottir et al., 2011).
Milk consumption is also a risk factor for prostate cancer as shown by meta-analysis of case-control studies (Qin et al, 2004). Milk intake in early life particularly promotes increased risk of advanced prostate cancer (Torfadottir et al, 2011). These authors investigated whether early-life residency in certain areas of Iceland marked by distinct differences in milk intake was associated with risk of prostate cancer in a population-based cohort of 8,894 men born between 1907 and 1935. Daily milk consumption in adolescence (vs. less than daily), but not in midlife or currently, was associated with a 3.2-fold risk of advanced prostate cancer. These data suggest that frequent milk intake in adolescence increases risk of advanced prostate cancer. In “The experience of Japan as a clue to the etiology of testicular and prostatic cancers” authors Ganmaa et al. (2003) show that the intake of milk, meat, and eggs increased 20, 9, and 7 fold, respectively, in Japan after world war 2. To examine the experience of Japan as a clue to the etiology, trends in the mortality rates of testicular and prostatic cancers from 1947 to 1998 were related to changes in dietary practices. The male population born before 1945 had a peak in death from testicular cancer in their thirties or forties, whereas those born after 1946 had a peak in their twenties. The death rate of prostatic cancer increased 25-fold almost linearly after the war. For the authors, in connection with the development and growth of testicular and prostatic cancers in Japan, particular attention should be paid to milk, because the increase in its consumption in the country is a recent occurrence and because milk contains considerable amounts of estrogens plus saturated fats.
Giovannucci et al. (1998) conducted a prospective study among 47,781 men of the “Health Professionals Follow-Up Study” free of cancer to examine whether calcium and fructose intake influenced risk of prostate cancer. Their findings provide indirect evidence for a protective influence of high calcitriol or vitamin D levels on prostate cancer and support increased fruit consumption and avoidance of high calcium intake to reduce the risk of advanced prostate cancer. Additional studies like the one by Ornish et al. (2005) show that intensive lifestyle changes may affect the progression of prostate cancer, including a vegan diet supplemented with soy (1 daily serving of tofu plus 58 gm of a fortified soy protein powdered beverage), fish oil (3 gm daily), vitamin E (400 IU daily), selenium (200 mcg daily) and vitamin C (2 gm daily), moderate aerobic exercise (walking 30 minutes 6 days weekly), stress management techniques (gentle yoga based stretching, breathing, meditation, imagery and progressive relaxation for a total of 60 minutes daily) and participation in a 1-hour support group once weekly to enhance adherence to the diet. Changes in serum PSA and LNCaP cell growth from baseline to 12 months were significantly different between the groups, showing more favorable changes in the experimental group, favoring a reduced risk in prostate cancer.
Additionally, there is the whole issue of calcium intake through milk to promote bone health. This has been increasingly found to be wrong, as milk consumption during teenage years is associated with hip fractures in older adults for example. Following Feskanich, Bischoff-Ferrari, Frazier and Willett (2014), over follow-up, 1226 hip fractures were identified in women and 490 in men. After controlling for known risk factors and current milk consumption, each additional glass of milk per day during teenage years was associated with a significant 9% higher risk of hip fracture in men, although teenage milk consumption was not associated with hip fractures in women.
Diet seems to be also an important factor in psychological health, where a vegan diet provides a protective effect. Tsai, Chang and Chi (2012) for example, found that more frequent consumption of vegetables seems to be protective against depressive symptoms in the elderly. It is worth noting that fruit, vegetable and antioxidant intakes are lower in older adults with depression as Payne, Steck, George and Steffens (2012) point out in their study. And grater than any other source, fruits and vegetables have one of the highest amounts of antioxidants, far more than animal products. As Carlsen et al. (2010) found, there are several thousand-fold differences in antioxidant content of foods. Spices, herbs and supplements include the most antioxidant rich products in their study, some exceptionally high. Berries, fruits, nuts, chocolate, vegetables and products thereof constitute common foods and beverages with high antioxidant values. This is relevant when people ask how one is going to substitute milk or meat when adopting a complete plant-based diet, this is like a meat-eating logic, the real question should be how one is going to substitute the antioxidants and nutrients present in plants but not in animal foods, one can substitute the proteins in meat with plant proteins, but one cannot do the same with antioxidants and nutrients found in plants, if we eat the proteins in plants we are also eating antioxidants and multiple nutrients, in addition to avoiding saturated fat and cholesterol for example. Given this information, a common myth that people raise against consuming vegetables and fruits is that they have a lot of pollutants from pesticides for example, it is worth mentioning in this case, that the vast majority of organic pollutants that are consumed through diet are argued to come from animal products, particularly animal fat (Lee at al., 2006; Magliano, Loh, Harding, Botton, & Shaw, 2014).
But going back to mood and psychological health, additional studies like the one by Akbaraly et al. (2009) confirm that dietary patterns have an impact in depressive symptoms. These authors found that after adjusting for potential confounders, participants in the highest tertile of the whole food pattern had lower odds of CES–D depression (Epidemiological Studies–depression Scale)than those in the lowest tertile. In contrast, high consumption of processed food was associated with an increased odds of CES–D depression. The authors conclude that in middle-aged participants, a processed food dietary pattern is a risk factor for CES–D depression 5 years later, whereas a whole food pattern is protective.
If we go back to the issue of Omega-6/Omega 3 ratio and its role on disease, authors Vaz, Kac, Nardi, and Hibbeln (2014) estimated the prevalence of suicide risk (SR) and major depressive episode (MDE) in early pregnancy, and the relationship of fatty acids serum status to these outcomes, and it was found that higher serum status of AA and ADA, two omega-6 fatty acids, were associated with greater likelihood of SR and MDE among pregnant Brazilian women independently of confounding variables.
Additionally, a vegan diet has the potential of increasing productivity and quality of life, including the aforementioned mental health. Katcher, Ferdowsian, Hoover, Cohen and Barnard (2010) determined the acceptability of a worksite vegan nutrition program and its effects on health-related quality of life and work productivity, where the vegan group reported improvements in general health, physical functioning, mental health, vitality and overall diet satisfaction compared with the control group. The vegan group also reported a decrease in food costs, and increased difficulty finding foods when eating out compared with the control group. The vegan group reported a 40–46% decrease in health-related productivity impairments at work and in regular daily activities.
How much can this whole food plant-base diet add in terms of years to our life span? Following Fraser and Shavlik (2001), and as we previously saw, California Adventists have higher life expectancies, they have higher ones at the age of 30 years than other white Californians by 7.28 years (95% confidence interval, 6.59-7.97 years) in men and by 4.42 years (95% confidence interval, 3.96-4.88 years) in women, giving them perhaps the highest life expectancy of any formally described population. As the authors mention, commonly observed combinations of diet, exercise, body mass index, past smoking habits, and hormone replacement therapy (in women) can account for differences of up to 10 years of life expectancy among Adventists. Also, healthy populations like the Adventists and Okinawans have high rates of centenarians. Not only do these populations live longer, they are healthier at higher age, not experiencing common age related illnesses, maintaining high mobility and mental capacity, as the previous studies show.
As a culmination to this essay, I leave here a link to doctor Greger’s talk “How Not To Die: The Role of Diet in Preventing, Arresting, and Reversing Our Top 15 Killers“, which covers many of the aspects here discussed and others.
References
Akbaraly, T. N., Brunner, E. J., Ferrie, J. E., Marmot, M. G., Kivimaki, M., & Singh-Manoux, A. (2009). Dietary pattern and depressive symptoms in middle age. The British Journal of Psychiatry, 195(5), 408-413.
Allen, N. E., Appleby, P. N., Davey, G. K., Kaaks, R., Rinaldi, S., & Key, T. J. (2002). The associations of diet with serum insulin-like growth factor I and its main binding proteins in 292 women meat-eaters, vegetarians, and vegans. Cancer Epidemiology and Prevention Biomarkers, 11(11), 1441-1448.
Allen, N. E., Appleby, P. N., Davey, G. K., & Key, T. J. (2000). Hormones and diet: low insulin-like growth factor-I but normal bioavailable androgens in vegan men. British Journal of Cancer, 83(1), 95.
Anderson, J. J., Darwis, N. D., Mackay, D. F., Celis-Morales, C. A., Lyall, D. M., Sattar, N., … & Pell, J. P. (2018). Red and processed meat consumption and breast cancer: UK Biobank cohort study and meta-analysis. European Journal of Cancer, 90, 73-82.
Appleton, B. S., & Campbell, T. C. (1983). Effect of high and low dietary protein on the dosing and postdosing periods of aflatoxin B1-induced hepatic preneoplastic lesion development in the rat. Cancer research, 43(5), 2150-2154.
Apter, D. A. N., & VIHKO, R. (1983). Early menarche, a risk factor for breast cancer, indicates early onset of ovulatory cycles. The Journal of Clinical Endocrinology & Metabolism, 57(1), 82-86.
Barnard, N. D., Cohen, J., Jenkins, D. J., Turner-McGrievy, G., Gloede, L., Green, A., & Ferdowsian, H. (2009). A low-fat vegan diet and a conventional diabetes diet in the treatment of type 2 diabetes: a randomized, controlled, 74-wk clinical trial–. The American journal of clinical nutrition, 89(5), 1588S-1596S.
Barnard, N. D., Cohen, J., Jenkins, D. J., Turner-McGrievy, G., Gloede, L., Jaster, B., … & Talpers, S. (2006). A low-fat vegan diet improves glycemic control and cardiovascular risk factors in a randomized clinical trial in individuals with type 2 diabetes. Diabetes care, 29(8), 1777-1783.
Barnard, R. J., Hong Gonzalez, J., Liva, M. E., & Ngo, T. H. (2006). Effects of a low-fat, high-fiber diet and exercise program on breast cancer risk factors in vivo and tumor cell growth and apoptosis in vitro. Nutrition and cancer, 55(1), 28-34.
Barnard, R. J., Hong, J., & Liva, T. H. N. (2005). Effect ofa Low-Fat, High-Fiber Diet and Exercise Intervention on Breast Cancer Risk Factors and Tumor Cell Growth & Apoptosis.
Bastide, N., Chenni, F., Audebert, M., Santarelli, R., Taché, S., Naud, N., … & Kuhnle, G. G. (2015). A central role for heme iron in colon carcinogenesis associated with red meat intake. Cancer research, canres-2554.
Brinkworth, G. D., Buckley, J. D., Noakes, M., Clifton, P. M., & Wilson, C. J. (2009). Long-term effects of a very low-carbohydrate diet and a low-fat diet on mood and cognitive function. Archives of internal medicine, 169(20), 1873-1880.
Carlsen, M. H., Halvorsen, B. L., Holte, K., Bøhn, S. K., Dragland, S., Sampson, L., … & Barikmo, I. (2010). The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutrition journal, 9(1), 3.
Darmadi-Blackberry, I., Wahlqvist, M. L., Kouris-Blazos, A., Steen, B., Lukito, W., Horie, Y., & Horie, K. (2004). Legumes: the most important dietary predictor of survival in older people of different ethnicities. Asia Pacific Journal of Clinical Nutrition, 13(2), 217-220.
De Biase, S. G., Fernandes, S. F. C., Gianini, R. J., & Duarte, J. L. G. (2007). Vegetarian diet and cholesterol and triglycerides levels. Arquivos brasileiros de cardiologia, 88(1), 35-39.
De la Torre, J. C. (2002). Vascular basis of Alzheimer’s pathogenesis. Annals of the New York Academy of sciences, 977(1), 196-215.
Dinu, M., Abbate, R., Gensini, G. F., Casini, A., & Sofi, F. (2017). Vegetarian, vegan diets and multiple health outcomes: a systematic review with meta-analysis of observational studies. Critical reviews in food science and nutrition, 57(17), 3640-3649.
Esselstyn, J. C., Gendy, G., Doyle, J., Golubic, M., & Roizen, M. F. (2014). A way to reverse CAD?. The Journal of family practice, 63(7), 356-364b.
Etemadi, A., Sinha, R., Ward, M. H., Graubard, B. I., Inoue-Choi, M., Dawsey, S. M., & Abnet, C. C. (2017). Mortality from different causes associated with meat, heme iron, nitrates, and nitrites in the NIH-AARP Diet and Health Study: population based cohort study. bmj, 357, j1957.
Farooqui, A. A., Horrocks, L. A., & Farooqui, T. (2007). Modulation of inflammation in brain: a matter of fat. Journal of neurochemistry, 101(3), 577-599.
Feskanich, D., Bischoff-Ferrari, H. A., Frazier, A. L., & Willett, W. C. (2014). Milk consumption during teenage years and risk of hip fractures in older adults. JAMA pediatrics, 168(1), 54-60.
Fraser, G. E., & Shavlik, D. J. (2001). Ten years of life: is it a matter of choice?. Archives of Internal Medicine, 161(13), 1645-1652.
Ganmaa, D., Li, X. M., Qin, L. Q., Wang, P. Y., Takeda, M., & Sato, A. (2003). The experience of Japan as a clue to the etiology of testicular and prostatic cancers. Medical hypotheses, 60(5), 724-730.
Gerster, H. (1998). Can adults adequately convert a-linolenic acid (18: 3n-3) to eicosapentaenoic acid (20: 5n-3) and docosahexaenoic acid (22: 6n-3)?. International Journal for Vitamin and Nutrition Research, 68(3), 159-173.
Giovannucci, E., Rimm, E. B., Wolk, A., Ascherio, A., Stampfer, M. J., Colditz, G. A., & Willett, W. C. (1998). Calcium and fructose intake in relation to risk of prostate cancer. Cancer Research, 58(3), 442-447.
Goff, L. M., Bell, J. D., So, P. W., Dornhorst, A., & Frost, G. S. (2005). Veganism and its relationship with insulin resistance and intramyocellular lipid. European journal of clinical nutrition, 59(2), 291.
Guevara-Aguirre, J., Balasubramanian, P., Guevara-Aguirre, M., Wei, M., Madia, F., Cheng, C. W., … & de Cabo, R. (2011). Growth hormone receptor deficiency is associated with a major reduction in pro-aging signaling, cancer, and diabetes in humans. Science translational medicine, 3(70), 70ra13-70ra13.
Hardy, K., Brand-Miller, J., Brown, K. D., Thomas, M. G., & Copeland, L. (2015). The importance of dietary carbohydrate in human evolution. The Quarterly review of biology, 90(3), 251-268.
Hunter, P. (2012). The inflammation theory of disease: The growing realization that chronic inflammation is crucial in many diseases opens new avenues for treatment. EMBO reports, 13(11), 968-970.
Hyson, D., Rutledge, J. C., & Berglund, L. (2003). Postprandial lipemia and cardiovascular disease. Current atherosclerosis reports, 5(6), 437-444.
Katcher, H. I., Ferdowsian, H. R., Hoover, V. J., Cohen, J. L., & Barnard, N. D. (2010). A worksite vegan nutrition program is well-accepted and improves health-related quality of life and work productivity. Annals of Nutrition and Metabolism, 56(4), 245-252.
Key, T. J., Appleby, P. N., Spencer, E. A., Travis, R. C., Roddam, A. W., & Allen, N. E. (2009). Cancer incidence in vegetarians: results from the European Prospective Investigation into Cancer and Nutrition (EPIC-Oxford)–. The American journal of clinical nutrition, 89(5), 1620S-1626S.
Le, L. T., & Sabaté, J. (2014). Beyond meatless, the health effects of vegan diets: findings from the Adventist cohorts. Nutrients, 6(6), 2131-2147.
Lee, D. H., Lee, I. K., Song, K., Steffes, M., Toscano, W., Baker, B. A., & Jacobs, D. R. (2006). A strong dose-response relation between serum concentrations of persistent organic pollutants and diabetes: results from the National Health and Examination Survey 1999–2002. Diabetes care, 29(7), 1638-1644.
Levy, Y., Maor, I., Presser, D., & Aviram, M. (1996). Consumption of eggs with meals increases the susceptibility of human plasma and low-density lipoprotein to lipid peroxidation. Annals of nutrition and metabolism, 40(5), 243-251.
Li, Y., Zhou, C., Zhou, X., & Li, L. (2013). Egg consumption and risk of cardiovascular diseases and diabetes: a meta-analysis. Atherosclerosis, 229(2), 524-530.
Lichtenstein, A. H., Ausman, L. M., Carrasco, W., Jenner, J. L., Gualtieri, L. J., Goldin, B. R., … & Schaefer, E. J. (1993). Effects of canola, corn, and olive oils on fasting and postprandial plasma lipoproteins in humans as part of a National Cholesterol Education Program Step 2 diet. Arteriosclerosis, Thrombosis, and Vascular Biology, 13(10), 1533-1542.
Magliano, D. J., Loh, V. H. Y., Harding, J. L., Botton, J., & Shaw, J. E. (2014). Persistent organic pollutants and diabetes: a review of the epidemiological evidence. Diabetes & metabolism, 40(1), 1-14.
Maruyama, K., Oshima, T., & Ohyama, K. (2010). Exposure to exogenous estrogen through intake of commercial milk produced from pregnant cows. Pediatrics International, 52(1), 33-38.
Farlow, D. W., Xu, X., & Veenstra, T. D. (2010). Consumption of cow’s milk and possible risk of breast cancer. Breast Care, 5, 44-46.
McCarty, M. F. (2003). A low-fat, whole-food vegan diet, as well as other strategies that down-regulate IGF-I activity, may slow the human aging process. Medical hypotheses, 60(6), 784-792.
Melina, V., Craig, W., & Levin, S. (2016). Position of the Academy of Nutrition and Dietetics: vegetarian diets. Journal of the Academy of Nutrition and Dietetics, 116(12), 1970-1980.
Moore, S. C., Lee, I. M., Weiderpass, E., Campbell, P. T., Sampson, J. N., Kitahara, C. M., … & Adami, H. O. (2016). Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA internal medicine, 176(6), 816-825.
Noto, H., Goto, A., Tsujimoto, T., & Noda, M. (2013). Low-carbohydrate diets and all-cause mortality: a systematic review and meta-analysis of observational studies. PloS one, 8(1), e55030.
Ornish, D., Weidner, G., Fair, W. R., Marlin, R., Pettengill, E. B., Raisin, C. J., … & Aronson, W. J. (2005). Intensive lifestyle changes may affect the progression of prostate cancer. The Journal of urology, 174(3), 1065-1070.
Pan, A., Sun, Q., Bernstein, A. M., Manson, J. E., Willett, W. C., & Hu, F. B. (2013). Changes in red meat consumption and subsequent risk of type 2 diabetes mellitus: three cohorts of US men and women. JAMA internal medicine, 173(14), 1328-1335.
Payne, M. E., Steck, S. E., George, R. R., & Steffens, D. C. (2012). Fruit, vegetable, and antioxidant intakes are lower in older adults with depression. Journal of the Academy of Nutrition and Dietetics, 112(12), 2022-2027.
Qin, L. Q., Xu, J. Y., Wang, P. Y., Kaneko, T., Hoshi, K., & Sato, A. (2004). Milk consumption is a risk factor for prostate cancer: meta-analysis of case-control studies. Nutrition and cancer, 48(1), 22-27.
Ruby, M. B., & Heine, S. J. (2011). Meat, morals, and masculinity. Appetite, 56(2), 447-450.
Schwingshackl, L., & Hoffmann, G. (2013). Low-carbohydrate (LC) diets impair flow-mediated dilatation (FMD): evidence from a systematic review and meta-analysis. British Journal of Nutrition, 110(5), 969-970.
Simopoulos, A. P. (2002). The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomedicine & pharmacotherapy, 56(8), 365-379.
Snowdon, D. A., & Phillips, R. L. (1985). Does a vegetarian diet reduce the occurrence of diabetes?. American Journal of Public Health, 75(5), 507-512.
Spence, J. D., Jenkins, D. J., & Davignon, J. (2012). Egg yolk consumption and carotid plaque. Atherosclerosis, 224(2), 469-473.
Spencer, E. A., Appleby, P. N., Davey, G. K., & Key, T. J. (2003). Diet and body mass index in 38 000 EPIC-Oxford meat-eaters, fish-eaters, vegetarians and vegans. International journal of obesity, 27(6), 728.
Stamler, J., Wentworth, D., & Neaton, J. D. (1986). Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded?: findings in 356 222 primary screenees of the multiple risk factor intervention trial (mrfit). Jama, 256(20), 2823-2828.
Sutliffe, J. T., Wilson, L. D., de Heer, H. D., Foster, R. L., & Carnot, M. J. (2015). C-reactive protein response to a vegan lifestyle intervention. Complementary therapies in medicine, 23(1), 32-37.
Torfadottir, J. E., Steingrimsdottir, L., Mucci, L., Aspelund, T., Kasperzyk, J. L., Olafsson, O., … & Jonsson, E. (2011). Milk intake in early life and risk of advanced prostate cancer. American journal of epidemiology, 175(2), 144-153.
Trumbo, P. R., & Shimakawa, T. (2011). Tolerable upper intake levels for trans fat, saturated fat, and cholesterol. Nutrition reviews, 69(5), 270-278.
Tsai, A. C., Chang, T. L., & Chi, S. H. (2012). Frequent consumption of vegetables predicts lower risk of depression in older Taiwanese–results of a prospective population-based study. Public Health Nutrition, 15(6), 1087-1092.
Vaz, J. S., Kac, G., Nardi, A. E., & Hibbeln, J. R. (2014). Omega-6 fatty acids and greater likelihood of suicide risk and major depression in early pregnancy. Journal of affective disorders, 152, 76-82.
Voight, B. F., Peloso, G. M., Orho-Melander, M., Frikke-Schmidt, R., Barbalic, M., Jensen, M. K., … & Schunkert, H. (2012). Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. The Lancet, 380(9841), 572-580.
Wang, Y., & Beydoun, M. A. (2009). Meat consumption is associated with obesity and central obesity among US adults. International Journal of Obesity, 33(6), 621.
Wiley, A. S. (2011). Milk intake and total dairy consumption: associations with early menarche in NHANES 1999-2004. PLoS One, 6(2), e14685.
Willcox, D. C., Scapagnini, G., & Willcox, B. J. (2014). Healthy aging diets other than the Mediterranean: a focus on the Okinawan diet. Mechanisms of ageing and development, 136, 148-162.
Wright, N., Wilson, L., Smith, M., Duncan, B., & McHugh, P. (2017). The BROAD study: A randomised controlled trial using a whole food plant-based diet in the community for obesity, ischaemic heart disease or diabetes. Nutrition & diabetes, 7(3), e256.
Yamamoto, S., Sobue, T., Kobayashi, M., Sasaki, S., & Tsugane, S. (2003). Soy, isoflavones, and breast cancer risk in Japan. Journal of the national cancer institute, 95(12), 906-913.
Yi, T., Li, S. M., Fan, J. Y., Fan, L. L., Zhang, Z. F., Luo, P., … & Chen, H. B. (2014). Comparative analysis of EPA and DHA in fish oil nutritional capsules by GC-MS. Lipids in health and disease, 13(1), 190.
Discover more from Sinapticas
Subscribe to get the latest posts sent to your email.
