©
Leandro Castelluccio
Image taken from link
Using neuroscience to explain cross-cultural differences is a relatively new trend, but it has grown enough to also fuel new distinct fields, such as cultural neuroscience, which investigates how human brain functions are shaped by interactions between culture, the brain and genes. This is an area where cultural psychology, social cognitive neuroscience, the study of neuroplasticity, and the study of culture-gene interactions come together (Han, Northoff, Vogeley, Wexler, Kitayama and Varnum, 2013). It has important advantages, like presenting an expanded toolkit of methodologies that different areas, like cognitive neuroscience and neurogenetics, are able to provide to allow a better understanding of culture (Ng, Morris and Oishi, 2013).
The way the above question is portrayed seems to be biased in assuming a causal relationship between brain function and culture, in the sense that culture can be explained on the basis of how our brain works, and by extension we would say that differences in brain function may explain cultural differences. We could adopt this perspective, but reality seems to be more complex than this. Research shows that there is a fine, bidirectional interaction, between genes, culture and brain function (Han et al., 2013). Actually, one of the challenges of cultural neuroscience is to distinguish culture-gene coevolution from gene-dominated generation of cultural values (Ng et al., 2013). The general accepted model is one in which the environment induces some pressure that affects the expression of certain genes and also certain cultural values, this comes together when we try to explain brain function, and the final outcome: behaviour (Chiao, Cheon, Pornpattananangkul, Mrazek and Blizinsky, 2013).
We need to be able to tell, therefore, the distinction between how cultural differences might be explained in terms of general (population) variance in brain function, and the fact that there are cultural differences in brain function, as it will be pointed out later on. Are cultural values shaping those differences in brain function, or is it the other way around? As stated before, the general conclusion is that there is a bidirectional interaction. If we do not come to an agreement on that matter, at least we can show how cultural differences correlate with differences in brain function, so either way, I think neuroscience can definitely tell us about cross-cultural differences.
There are many factors within culture that shapes brain processes, in fact, another challenge of the field of cultural neuroscience is about addressing within-nation variation and socio-ecological factors (Ng et al., 2013). It has been found, for example, that people with high socio-economic status are more likely to show increased neural response in the ventral striatum when perceiving high-status information about others. People with low socio-economic status show increased ventral striatum activation when perceiving low-status information (Chiao et al., 2013). Another example is racial identification. It has been found that people’s racial identification modulates neural response within cortical midline structures when empathizing the pain of other members of their own racial group (Chiao et al., 2013). These areas appear to be involved in self-referential processing (Northoff, Heinzel, De Greck, Bermpohl, Dobrowolny and Panksepp, 2006). We could name other examples, but the idea is that it should be expected that culture plays a fundamental role in shaping brain function, and differences in cultural values would imply differences in brain processes.
We could say that the dimensions we use to analyse culture are the same, but they differ across populations, and that would be a cross-cultural difference. Let’s take the example of religion. This is a widespread phenomenon, but there might be individual differences in the way people process it, thus creating a cross-cultural difference. Evidence shows that Koreans who demonstrated greater religiosity and are homozygous for the G allele of the OXTR receptor polymorphism r253576 (oxytocin receptor) were more prone to report greater psychological well-being. However, Caucasian Americans showing grater religiosity and carrying the same genetic variation of this receptor, demonstrated reduced psychological well-being (Chiao at al., 2013). This difference could further alter the way religion is perceived and manifested in a particular culture. This is a way of interpreting cross-cultural differences where individual differences, that in this case affect neural chemistry, could shape cultural values and phenomena.
Some of the early studies showing how individual differences could shape distinct cultural values looked at the genetic variation of the serotonin transporter (5-HTTLPR) and its influence on the individualistic vs collectivistic cultural framework (Way and Lieberman, 2010). Although we are addressing a genetic variation, like we mentioned before, its impact is focused on the brain and how it responds to stimuli, and in this case in particular: social stimuli. As Way and Liberman (2010) mention, short/short variation of 5-HTTLPR, MAOA-uVNTR (variation of the monoamine oxidase gene) and also OPRM1 A118G (variation of the m-opioid receptor gene) have effects on social sensitivity, i.e. the degree of emotional responsiveness to social events and experiences. In the case of the serotonin transporter, it is argued that the broad cultural differences in interdependent vs independent modes of self-construal in Asian vs Western cultures may have arisen, in part, because of the difference in the frequency of homozygosity for the short allele of the transporter’s gene. Short allele increases an individual’s risk for depression following stressful events and Asian populations exhibit a higher frequency of this variance relative to Westerners (Ames and Fiske, 2010). Way and Liberman (2010) add that short-allele implies sensitivity to the depression-inducing effects of social stress. According to these authors, having a rich, interconnected social network, like the ones in collectivistic cultures, might help maintaining the well-being of those carrying the short/short variation. As they argue, individuals of East Asian descent living in the USA suffer higher levels of major depression, compared to Asians living in Asia, which goes in accordance with their interpretation.
Something similar is the case for the other genetic variations. MAOA-uVNTR was associated with the degree of exclusion-related neural activation. At the same time, it appears that the OPRM1 A118G variation is associated to self-reported dispositional sensitivity to rejection (Way and Liberman, 2010).
But how accurate is this causal model regarding cultural differences? When one looks at Japanese culture, for example, a country with a high frequency of the short/short variation of the serotonin transporter, it seems that it exhibits a much more westernized way of living. One could say that this is a recent event, and that originally Japan was more collectivistic, which is consistent with the frequency of the serotonin variation. One way to test for the model’s assumptions would be to study whether the rate of depression has increased as a function of the westernization of the Japanese culture (while controlling for other variables as far as possible). We could argue, on the other hand, that medieval Europe was much more collectivistic if we compare it to modern Europe, considering how the role of the individual was seen at that times, even though Western-European populations have a lesser frequency of short/short variation. At least one could conclude that these genetic differences are not enough to prevent such changes in cultural values. We could interpret, however, that it is not the fact that collectivistic cultures exert a sort of protection for short/short individuals, but rather, collectivistic cultures would tend to perpetuate and be more resistant to changes when individuals are more affected by things like social exclusion, i.e. if one is less eager to challenge social institutions and their organization, the present cultural values would expand and remain within the population. This would be a case where culture (the prevalent values in the society) is selecting certain kinds of genes and behaviour. For example, Mrazek, Chiao, Blizinsky, Lun and Gelfand (2013) investigated the coevolution of cultural-tightness and the serotonin transporter gene in the production of moral justifiability, depending on variations in ecological threat. As they indicate, it seems that tight nations are more likely to endorse strict adherence to social norms, while loose nations are more likely to be tolerant of violations of social norms. Tightness refers to the sensitivity of social norm compliance while looseness refers to social norms violations. They found across 21 nations, that ecological threat predicts cultural tightness–looseness due to the frequency of short/short variation, and that frequency of S allele variation predicts the justifiability of moral behaviour based on cultural tightness-looseness. This could be interpreted as an indication that adaptive moral behaviour results from both cultural and genetic selection of tightness-looseness and the serotonin transporter gene (Mrazek et al., 2013). So the direction of causation could go in either way. As these authors consider, ecological and human made threats increase the selection for S allele carriers, as these individuals are more likely to detect such environmental threats and avoid them. At the same time, because of the heightened sensitivity to threats, S allele individuals are also more inclined to develop strong norms that help them to coordinate actions to deal with these threats.
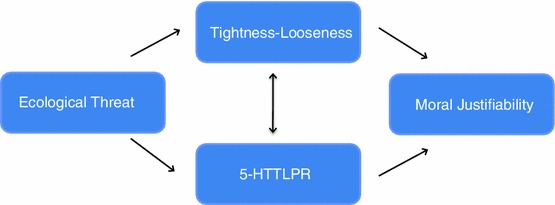
(Figure taken from Mrazek et al., 2013)
The relationship between culture and genetics and brain function may be even more complicated. It has been found, for example, that people from collectivistic cultures are more likely to prefer expressive (emotional) suppression, while people from individualistic culture are more likely to prefer cognitive reappraisal, and this seems to be related to genetics differences for the OXTR (Chiao et al., 2013). According to these authors, A allele carries in Korea are more likely to prefer emotional suppression compared to G allele carries, but interestingly, this pattern is the opposite in Americans, which suggests that culture interacts with the OXTR in preference for emotion regulation strategy. One way of interpreting this is that not only culture may affect which genes are more prevalent, but the kind of behaviour the genes will influence.
We could also argue that collectivistic and individualistic cultures are not just about the relationship between the person and the group, there are differences in cognitive processes in both as well. One might say that collectivistic and individualistic cultures could have arisen as a function of differences in these cognitive processes, rather than a way to protect for vulnerabilities. For example, evidence shows that East Asians and Westerners have different perceptual styles in decoding visual scenes. Westerners tend to focus on objects in an analytical way, disregarding the context, while East Asians tend to do the opposite, focusing more on context (Nisbett & Miyamoto, 2005). In accordance with this, people living in the United States and Japan who endorse individualistic cultural values for example, have more frequently increased neural activity in medial prefrontal cortex to general statements compared to contextual self-statements, while people who endorse collectivistic cultural values show the opposite: increased medial prefrontal cortex response to contextual compared to general self-statements. Additionally, it has been found that cultural priming modulates neural response within dorsal, but not ventral, portions of the medial prefrontal cortex, when people are shown autobiographical facts. These results suggests that cultural values modulate neural response during self-processing (Chiao et al., 2013). Another example of culture modulating neural responses can be found in the research about individualism-collectivism and the neural bases of emotion. Evidence suggests that people living in a collectivistic culture, like Japan, show increased amygdala response when compared to people living in an individualistic culture, like the United States. This effect appears to be independent of other factors modulating amygdala response, like genotype, gender, personality or urbanity. At the same time, it has been found that for Asians living in Europe, amygdala response to emotions expressed by Caucasian Europeans depends on length of stay in Europe (Chiao et al., 2013). We can say, based on all this, that cultural values modulate neural activity, but we also have evidence of differences in cognitive processes that might impact the prevalence of cultural values within a population.
It appears that there is greater object-focused processing among Westerners. For example American participants have demonstrated stronger and more distributed neural activations during object processing in areas such as the middle temporal gyrus, right superior temporal-supramarginal gyrus and superior parietal lobule. But few cross-cultural brain differences were associated with background processing, therefore, it is thought, contrary to the idea that there is greater attention to context among East Asians, that these differences result from additional object processing among Westerners (Ames and Fiske, 2010). This difference might have emerge as a result of cultural immersion, as research shows that neural differences between groups of Asians and Westerners correlates with the number of years during which those groups have had divergent experiences, that is, object processing in Americans versus Singaporeans, for example, emerged only for older adults, and not for younger participants (Ames and Fiske, 2010). This suggests that it is culture affecting brain function rather than the other way around. This effect has been controlled for possible influences by developmental, neurobiological, and genetic factors that might have contributed to its appearance, for example, in one experiment, priming participants with a Western, independent self-construal style, led to greater activation of the lateral occipital cortex, an area related to object processing, during a task involving local versus global visual targets (Ames and Fiske, 2010). Following the above it has been found that the recruitment of attentional resources varies among certain task for different cultures, in this case less attentional processing is required for culturally preferred modes (Ames and Fiske, 2010).
Also, research shows that neural activation related to basic numerical processes varies considerably across cultures (Han and Northoff, 2008). At the same time, neural activation in case of language processing is different, for example, native Chinese speakers activate dorsal regions of the inferior parietal lobe when reading Chinese characters, while native English speakers recruit the superior temporal gyrus for reading English words (Han and Northoff, 2008).
There is a limit, however, to the extent in which differences in culture could generate differences in brain function. Park and Gutchess (2006) looked at aging and cross-cultural differences. They show a distinction between cognitive processes that decline in a similar way across cultures, indicating the universality of cognitive aging, and aging that is different among cultures, as it is expressed, for example, by differences in activation of object- processing areas in old adults from Asian versus Western cultures. Han and Northoff (2008) talk in a similar fashion about the findings which shows a distinction between culture-sensitive and culture-invariant neural mechanisms of human cognition.
Other dimensions in which cultures vary from each other have been addressed in terms of differences in gene variation related to brain function. For example, the Serotonin Transporter Gene (SLC6A4) has been studied in relation to dominance and power. Social dominance behaviour of populations in high power distance index (PDI) cultures appears to be affected by the frequency of S-allele carriers compared to L-allele carriers. The former seem to be more sensitive to social cues. Evidence shows that rhesus monkey species, for example, that have higher tolerant societies with less hierarchy and relaxed dominance tend to carry only the L-allele (Chiao et al., 2013). Preference for social hierarchy itself is something that seems to be shaped by cultural values. It has been found that people from places like Korea, a nation argued to prefer social hierarchy, show increased empathic neural response within the left temporoparietal junction, when viewing people from their own culture in pain, compared to neutral scenarios and when compared to people living in the United States, for example, a nation that prefers egalitarianism. This seems to be related to the recruitment of brain regions associated with theory of mind (like the temporoparietal junction) when responding to the pain of others in a hierarchical culture (Chiao et al., 2013). As mentioned by these authors, it is argued that the activation of this area is not as important in more egalitarian cultures, because they allow more emotional expression compared to high hierarchical cultures, which helps increase social cues that allows people to interpret and respond to other people’s feelings when in painful or neutral situations.
There are still challenges in using neuroscience to understand cross-cultural differences, one of the most relevant refers to economic barriers, like the ones regarding the tremendous cost of MRI scanners and training personnel. There is also a need for bridging diverse research within cultural neuroscience (Ng, Morris and Oishi, 2013). This, I consider, would be important for the progress of our understanding of cross-cultural differences. But still, I think neuroscience can tell us a lot about these cross-cultural differences. We looked and criticized the evidence showing the possible relationships between genes, brain function and culture, and although many aspects remain unclear, we can conclude that there are important interactions between these aspects, but we need to keep asking questions about the exact relationship between these elements.
References
Ames, D., & Fiske, S. (2010). Cultural neuroscience. Asian Journal of Social Psychology, 13(2), 72-82.
Chiao, J., Cheon, B., Pornpattananangkul, N., Mrazek, A., & Blizinsky, K. (2013). Cultural Neuroscience: Progress and Promise. Psychological Inquiry, 24(1), 1-19.
Han, S., Northoff, G., Vogeley, K., Wexler, B., Kitayama, S., & Varnum, M. (2013). A Cultural Neuroscience Approach to the Biosocial Nature of the Human Brain. Annual Review of Psychology, 64, 335-359.
Han, S., & Northoff, G. (2008). Culture-sensitive neural substrates of human cognition: A transcultural neuroimaging approach. Nature Reviews Neuroscience, 9(8), 646-654.
Mrazek, A. J., Chiao, J. Y., Blizinsky, K. D., Lun, J., & Gelfand, M. J. (2013). The role of culture–gene coevolution in morality judgment: examining the interplay between tightness–looseness and allelic variation of the serotonin transporter gene. Culture and Brain, 1(2-4), 100–117. http://doi.org/10.1007/s40167-013-0009-x
Ng, B., Morris, J., & Oishi, S. (2013). Cultural Neuroscience: The Current State of Affairs. Psychological Inquiry, 24(1), 53-57.
Nisbett, R. E. & Miyamoto, Y. (2005). The influence of culture: Holistic versus analytic perception. Trends in Cognitive Sciences, 9 (10), 467–473.
Northoff, Georg, Heinzel, Alexander, De Greck, Moritz, Bermpohl, Felix, Dobrowolny, Henrik, & Panksepp, Jaak. (2006). Self-referential processing in our brain—A meta-analysis of imaging studies on the self. Neuroimage, 31(1), 440-457.
Park, D., & Gutchess, A. (2006). The cognitive neuroscience of aging and culture. Current directions in psychological science, 15(3), 105-108.
Way, B. M., & Lieberman, M. D. (2010). Is there a genetic contribution to cultural differences? Collectivism, individualism and genetic markers of social sensitivity. Social cognitive and affective neuroscience, 5(2-3), 203-211.
Discover more from Sinapticas
Subscribe to get the latest posts sent to your email.

2 thoughts on “Can neuroscience tell us anything about cross-cultural differences?”